Abstract
Growing evidence links air pollution, a ubiquitous environmental stressor, to a higher risk of developing mental disorders, raising significant public health concerns. Mental disorders represent a significant global public health challenge which can have a profound impact on individual lives. In this study, we used Mendelian randomization (MR) to investigate the causal relationship between ambient air pollution and four common mental disorders. Genome-wide association study (GWAS) data for ambient air pollution and summary-level GWAS data for four representative mental disorders were obtained from open-access database. Inverse variance weighted (IVW) method with multiplicative random-effects model was the main analysis. Sensitivity analyses were conducted to validate the results. Bayesian colocalization analysis was conducted to explore the potential shared genetic causal variants between specific air pollutants and mental disorders. A suggestive association was observed between political matter (PM) 2.5 and anxiety disorders (OR 2.96, 95%CI 1.29–6.81, p = 0.010). Exposure to nitrogen dioxide (NO2) was significantly linked to an elevated risk of schizophrenia (OR 1.95, 95% CI 1.45–2.63, p = 1.13E-05) and showed a nominal association with an increased risk of bipolar disorder (OR 1.43, 95% CI 1.09–1.86, p = 0.009). A suggestive causal association was detected between nitrogen oxides (NOx) and anxiety disorder (OR 2.90, 95%CI 1.21–6.97, p = 0.017). No significant association was detected between exposure to PM2.5-10, PM10 and mental disorders. No significant horizonal pleiotropy and heterogeneity was found. The colocalization analysis revealed robust evidence supporting the colocalization of NO2 with schizophrenia at SNP rs12203592. Our findings support causal associations between exposure to ambient air pollution, particularly PM2.5, NO2, and NOx, and an increased risk of specific mental disorders.
Similar content being viewed by others
Highlights
-
1.
Mendelian randomization was first applied to the field of air pollution to investigate its impact on mental disorders.
-
2.
The large sample size from public GWAS databases through strict screening contributed to the compelling results.
-
3.
PM2.5, NO2, and NOx exhibit causal associations with specific mental disorders.
Introduction
Ambient air pollution is a major environmental health risk, estimated to cause 7 million premature deaths annually worldwide [1]. It is a complex mixture of various pollutants emitted from natural and anthropogenic sources, including vehicles, power plants, industrial facilities, and wildfires. These pollutants can have significant adverse effects on human health, especially the respiratory and cardiovascular systems [2,3,4]. Particulate matter (PM) is one of the most concerning air pollutants due to its ability to penetrate deep into the lungs and enter the bloodstream [5]. PM is classified by the size of the particles into coarse particles (<10 μm in size, PM10), fine particles (<2.5 μm in size, PM2.5), and ultrafine particles (<1 nm in size, PM0.1) [6]. Nitrogen oxides (NOx) are a group of gases that form when fuel is burned at high temperatures [7]. NOx can contribute to the formation of smog and acid rain, and can also irritate the respiratory system. Nitrogen dioxide (NO2), a specific type of nitrogen oxides (NOx), is an independent contributor to adverse health effects [8]. Other common air pollutants include sulfur dioxide, carbon monoxide, volatile organic compounds.
Mental disorders represent a significant global public health challenge, affecting hundreds of millions of individuals worldwide. According to the World Health Organization (WHO), mental disorders contribute to an estimated 13% of the global burden of disease [9]. These disorders can have a profound impact on individual lives, impairing functioning in work, school, and social relationships [10]. Moreover, mental disorders generate substantial societal and economic costs. Key epidemiological features of mental disorders include their high prevalence, affecting approximately one in four people globally [9]. While they can occur at any age, mental disorders exhibit a peak incidence in young adulthood [11]. Furthermore, they constitute a leading cause of disability and premature mortality. The global economic burden associated with mental disorders is estimated to be a staggering $1 trillion annually. Common mental disorders encompass a spectrum of conditions, including depression, anxiety disorders, and schizophrenia [12]. Efforts to address this global challenge necessitate increased public awareness about mental disorders, improved access to effective mental healthcare services, and a reduction in the stigma associated with mental illness.
Several studies have investigated the relationship between air pollution and mental disorders. A population-based prospective cohort study from UK Biobank indicated that PM2.5 and NOx were associated with increased risk of major depression disorder [13]. Another long-term prospective study in Italy found that an increase in PM2.5 was associated with a hazard ratio of 1.070 for schizophrenia spectrum disorder, 1.135 for depression, and 1.097 for anxiety disorders [14]. A time-series study in Shanghai suggested that short-term exposure to air pollution, particularly sulfur dioxide, was associated with an increased risk of hospital admissions for mental disorders [15]. A study conducted in Poland on children with attention deficit hyperactivity disorder demonstrated that exposure to air pollution, particularly short-term exposure to NO2, may negatively affect attention in children [16]. A large population-based cohort study of children born in Southern California indicated that maternal exposure to PM2.5 during pregnancy was independently associated with an increased risk of autism spectrum disorder [17]. Similarly, in Southern California, researchers found that long-term exposure to air pollution during the antepartum and postpartum periods was associated with an increased risk of postpartum depression [18].
Mendelian randomization (MR) has emerged as a powerful tool in epidemiology, leveraging genetic variants as instrumental variables to estimate the causal effects of environmental exposures on health outcomes [19, 20]. This approach offers distinct advantages over traditional observational studies. It is less susceptible to confounding bias due to the random allocation of genetic variants at conception. Additionally, MR studies can achieve greater statistical power by mitigating the influence of confounding factors. Furthermore, MR uniquely allows for the investigation of time-varying exposures, which are challenging to assess accurately in observational studies. This approach has the potential to yield novel insights into the association between these factors, informing future research endeavors. In this study, we used Mendelian randomization (MR) to investigate the causal relationship between ambient air pollution and four common mental disorders. As a complement of MR analysis, Bayesian colocalization analysis was conducted to explore the potential shared genetic causal variants between specific air pollutants and mental disorders.
Materials and methods
Study design
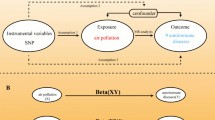
We conducted a two-sample MR study following the Strengthening the Reporting of Observational Studies in Epidemiology using Mendelian Randomization (STROBE-MR) guidelines [21]. Relevant single nucleotide polymorphisms (SNPs) were selected from the genome-wide association study (GWAS) summary results after implementing quality control procedures. We aimed to investigate the causal relationship between ambient air pollution and representative mental disorders using MR and relevant analyses. Three key assumptions were required to be met in MR: 1. IVs are significantly associated with the exposures; 2. IVs are not associated with the outcome through confounders; 3. IVs do not directly affect outcome.
The UK Biobank cohort study was approved by the North West Research Ethics Committee. All participants provided informed consent. The study was conducted in accordance with the Declaration of Helsinki. Ethical approval was not required for the analyses of summary statistics. This was because the analyses were based on anonymized data.
Data sources for exposure and outcome
This Mendelian randomization (MR) study was primarily conducted in individuals of European ancestry. Genome-wide association study (GWAS) data for ambient air pollution were obtained from the UK Biobank, a large-scale biomedical database and research resource [22]. The UK Biobank contains de-identified genetic, lifestyle, and health information, as well as biological samples, from over 500,000 United Kingdom participants. Five types of ambient air pollution were considered for further analyses: particulate matter with an aerodynamic diameter of ≤2.5 μm (PM2.5), particulate matter with an aerodynamic diameter of 2.5–10 μm (PM2.5–10), particulate matter with an aerodynamic diameter of ≤10 μm (PM10), nitrogen dioxide (NO2), and nitrogen oxides (NOx). The sample sizes were approximately 42,000 for PM2.5, PM2.5–10, and PM10, and approximately 45,000 for NO2 and NOx. A land use regression (LUR) model was employed to estimate the levels of air pollution at the residential addresses of the study participants.
Summary-level GWAS data for four representative mental disorders were obtained as outcomes from recent large-scale meta-analyses of GWAS conducted by the Psychiatric Genomics Consortium (PGC). Summary-level GWAS data for depression from a meta-analysis of 33 cohorts included 500,199 individuals (170,756 cases and 329,443 controls) with 8,483,301 variants analyzed [23]. Summary-level GWAS data for anxiety disorder from seven independent studies participating in the Anxiety Neuro Genetics Study (ANGST) were obtained. The dataset included 17,310 individuals and 6.5 million imputed SNPs [24]. The summary-level GWAS dataset for schizophrenia comprised a total of 36,989 cases and 113,075 controls [25]. Summary-level GWAS data for bipolar disorder were obtained from 57 datasets, comprising 41,917 cases and 371,549 controls [26].
Genetic instrumental variables selection
A p-value of less than 5e-08 is the most commonly used threshold for initial IV screening. However, in the present study, this threshold resulted in the identification of a limited number of IVs. Therefore, a more liberal threshold of p < 5e-06 was adopted.
To remove linkage disequilibrium (LD) between instrumental variables (IVs), a threshold of r2 < 0.001 and clump distance >10,000 kb (1000 genome reference panel) was applied. If an IV extracted from the exposure dataset did not match any SNPs in the outcome dataset, a proxy SNP in LD with r2 > 0.8 was used. SNPs without alternative alleles were excluded from the analysis. The F-statistic (F = Betaexposure2/SEexpore2) was applied to evaluate the strength of the IVs. IVs with an F-statistic < 10 were considered weak IVs and were excluded from the study. To further validate assumption 3, we excluded IVs that were potentially statistically significantly associated with the outcome (p < 5e-04). After harmonization, palindromic SNPs with ambiguous strands and ambiguous SNPs with unconcordant alleles were directly excluded. The IVs screened for MR analyses are presented in Supplementary Table 1.
Mendelian randomization and sensitivity analyses
Inverse variance weighted (IVW) method with multiplicative random-effects model was the main analysis to evaluate the causal relationship between air pollution and mental disorders. This method first estimates the causal effect of each single SNP on the outcome by calculating the Wald ratio [27]. It then uses the inverse variance of the SNPs as weights to obtain a combined causal effect. To further validate the primary results, we employed several supplementary methods, including the MR-Egger method, the weighted median method, the simple method, and the weighted model method. Among the five estimation methods, the IVW method had the highest statistical power. All five methods yielded consistent results in the same direction, indicating a reliable causal estimate.
We conducted sensitivity analyses to assess between-SNP heterogeneity and horizonal pleiotropy in the MR analyses. Heterogeneity was assessed using Cochran’s Q test with the inverse-variance weighted (IVW) method and the MR-Egger method. A p > 0.05 indicated no significant heterogeneity. To assess horizonal pleiotropy in the MR analyses, we employed both MR-Egger intercept test and the Mendelian randomization pleiotropy residual sum and outlier (MR-PRESSO) test. A p > 0.05 in the MR-Egger intercept test and MR-PRESSO global test indicated no significant horizonal pleiotropy. Additionally, MR-PRESSO can identify outliers among the IVs.
Mendelian randomization relies on the assumption of no pleiotropy. The presence of pleiotropy can bias MR estimates and lead to spurious associations. In our study, we evaluated pleiotropy after conducting the primary MR analyses. If significant pleiotropy was detected, we performed an iterative outlier exclusion approach. Briefly, we excluded IVs one-by-one in ascending order of their influence on the pleiotropy test statistic until a p-value greater than 0.05 was obtained. The MR analyses were then repeated with the remaining IVs.
To further verify the pleiotropic effects of air pollution on mental disorders, we used PhenoScanner V2 (http://www.phenoscanner.medschl.cam.ac.uk) to investigate whether the observed associations were confounded by potential factors such as smoking, pesticide use, and socioeconomic status [28]. We also conducted leave-one-out sensitivity analyses to evaluate the influence of potentially pleiotropic SNPs on the causal estimates. This was done by systematically removing one SNP at a time and re-estimating the causal effect. The two methods complemented the MR-PRESSO test to identify and exclude potential pleiotropic IVs.
Colocalization analysis
Based on summary GWAS data, we conducted Bayesian colocalization analysis to explore the potential shared genetic causal variants between specific air pollutants and mental disorders within a designated genomic region. For this purpose, we established a 1MB window around the lead SNP to pinpoint pertinent genomic areas. In summary, the colocalization analysis is grounded in five hypotheses: H0 posits no association with either trait; H1 indicates an association with trait 1 but not trait 2; H2 suggests an association with trait 2 but not trait 1; H3 proposes associations with both traits through two independent SNPs; and H4 asserts that both traits are associated through a single shared SNP [29]. As per established guidelines, a posterior probability of H4 (PP.H4) ≥ 80% is indicative of robust evidence for colocalization [30]. These findings suggest the existence of a common genetic variant linking specific air pollutants and mental disorders, thereby pointing to a possible shared genetic mechanism underlying this relationship.
Statistical analysis
All analyses in the current study were conducted using R software (version 4.2.3). Data processing was performed using the TwoSampleMR and MRPRESSO packages [31]. The MR estimates were resented as odds ratios (ORs) with 95% confidence intervals (CIs). Colocalization analysis was performed employing the Coloc package (https://github.com/chr1swallace/coloc) [32].
Considering the MR analysis with multiple outcomes, we used a Bonferroni-corrected significance threshold of P = 0.0025 (α = 0.05/(Nexposure*Noutcome)) at the individual outcome level in our primary analysis. This was to control for the family-wise error rate. MR estimates with a P-value less than 0.0025 were considered to be significantly causally associated, while a P-value between 0.0025 and 0.05 indicated suggestive evidence for an association. The significance thresholds were determined a priori.
Results
Main analyses
Figure 1 illustrated the comprehensive workflow of the present MR study. Figure 2 showed the results of the IVW method. Genetic liability to PM2.5 was significantly associated with an increased risk of schizophrenia (OR 1.79, 95%CI 1.25–2.56, p = 0.001). Suggestive associations were also identified between genetic susceptibility to PM2.5 and several psychiatric disorders: major depressive disorder (OR 1.18, 95% CI 1.01–1.38, p = 0.033), anxiety disorders (OR 2.96, 95% CI 1.29–6.81, p = 0.010), and bipolar disorder (OR 1.52, 95% CI 1.12–2.07, p = 0.007). Exposure to NO2 was significantly linked to an elevated risk of schizophrenia (OR 1.95, 95% CI 1.45–2.63, p = 1.13E-05) and showed a nominal association with an increased risk of bipolar disorder (OR 1.43, 95% CI 1.09–1.86, p = 0.009). Exposure to NOx was nominally associated with an increased risk of schizophrenia (OR 1.49, 95%CI 1.10–2.04, p = 0.011). Meanwhile, a potential causal association was identified between NOx exposure and anxiety disorders (OR 2.90, 95% CI 1.21–6.97, p = 0.017). No significant association was detected between exposure to PM2.5-10, PM10 and mental disorders.
To strengthen the findings from the IVW method, we further evaluated the robustness of the causal associations using four additional methods (presented in Supplementary Figs. 1–5) and scatterplots of MR estimates with positive IVW results (Fig. 3A–H). MR estimates for PM2.5 and depression, schizophrenia, and bipolar disorder (Fig. 3A, C, D, Supplementary Fig. 1), as well as NOx and schizophrenia (Fig. 3H, Supplementary Fig. 5), differed in direction from the IVW results, suggesting inconclusive causal associations for these exposures and outcomes. However, the causal associations identified by the IVW method for PM2.5 and anxiety disorder (Fig. 3B, Supplementary Fig. 1), NO2 and anxiety disorder (Fig. 3E, Supplementary Fig. 4), NO2 and schizophrenia (Fig. 3F, Supplementary Fig. 4), and NOx and anxiety disorder (Fig. 3G, Supplementary Fig. 5) were corroborated by the consistent direction of effect observed across all five MR estimates, suggesting robustness.
A PM2.5 on depression; B PM2.5 on anxiety disorder; C PM2.5 on schizophrenia; D PM2.5 on bipolar disorder; E NO2 on schizophrenia; F NO2 on bipolar disorder; G NOx on anxiety disorder; H NOx on schizophrenia. SNP single nucleotide polymorphism, PM particulate matter, NO2 nitrogen dioxide, NOx nitrogen oxide, MR mendelian randomization.
Sensitivity analyses
After excluding outliers based on our exclusion strategy, neither the MR-Egger intercept test nor the MR-PRESSO global test detected evidence of horizontal pleiotropy in the MR estimates. (Table 1). Similarly, the MR-Egger intercept test also yielded no evidence of horizontal pleiotropy (Table 1). Furthermore, no significant heterogeneity was detected between air pollution exposures and mental health outcomes (Table 1). Funnel plots constructed from positive MR estimates derived from MR-Egger regression displayed a symmetrical distribution of instrumental variables (IVs) in Supplementary Fig. 6, further supporting the absence of significant pleiotropy. Leave-one-out sensitivity analyses (Fig. 4A–H) conducted on the positive MR results did not reveal any influential outliers. Removing individual IVs one at a time did not significantly alter the pooled MR estimates.
A PM2.5 on depression; B PM2.5 on anxiety disorder; C PM2.5 on schizophrenia; D PM2.5 on bipolar disorder; E NO2 on schizophrenia; F NO2 on bipolar disorder; G NOx on anxiety disorder; H NOx on schizophrenia. SNP single nucleotide polymorphism, PM particulate matter, NO2 nitrogen dioxide, NOx nitrogen oxide, MR Mendelian randomization.
Colocalization analysis
Subsequently, we performed comprehensive Bayesian colocalization analyses to explore the potential shared causal variants linking air pollutants and mental disorders. The colocalization analysis revealed robust evidence supporting the colocalization of NO2 with schizophrenia at SNP rs12203592 (PP.H4 = 1.00), positioned on chromosome 6 within the interferon regulatory factor 4 (IRF4) gene region (Supplementary Table 2). SNP rs12203592 served as a significant IV (p = 3.20E-15 in the GWAS for NO2) (Supplementary Table 1) in the MR analysis designed to determine the causal relationship between NO2 and schizophrenia. The rest pairs of specific air pollutants and mental disorders exhibited limited evidence of colocalization.
Discussion
Given the well-documented detrimental effects of air pollution on human health, research is crucial to elucidate the specific air pollution challenges faced by different regions. The present study investigates the causal relationship between ambient air pollution and common mental disorders using the Mendelian randomization (MR) method. Our robust findings demonstrate significant adverse effects of NO2 exposure on schizophrenia (IVW, OR 1.95, 95% CI 1.45–2.63, p = 1.13E-05). Additionally, our study identified potential associations between PM2.5 exposure and anxiety disorders (IVW, OR 2.96, 95% CI 1.29–6.81, p = 0.010), NO2 exposure and bipolar disorders (IVW, OR 1.43, 95% CI 1.09–1.86, p = 0.009), as well as NOx exposure and anxiety disorders (IVW, OR 2.90, 95% CI 1.21–6.97, p = 0.017). However, the evidence for PM2.5’s influence on depression, schizophrenia, and bipolar disorder, and the link between NOx and schizophrenia requires further investigation. Notably, we found no causal association between PM2.5-10, PM10 exposure, and any of the investigated mental disorders. To further corroborate the findings from the MR analysis, we performed Bayesian colocalization analysis, which indicated that SNP rs12203592 colocalizes NO2 with schizophrenia.
Mendelian randomization and colocalization are two statistical methods utilized with summary data from GWAS to elucidate the connections between traits and diseases. Although they share a common purpose, their objectives, methodologies, and interpretations differ significantly due to their origins in distinct scientific fields. Mendelian randomization evaluates whether genetic predictors of an exposure correlate with an outcome, interpreting this association as evidence of a causal effect. In contrast, colocalization examines whether two traits are influenced by the same or different causal variants. The two approaches can offer complementary insights for identifying potential causal associations [33]. It has been documented that IRF4 rs12203592 is involved in pathway-specific risks associated with melanoma development [34, 35], as well as in the determination of skin freckling and eye color [36,37,38].
The well-established theory that air pollutants can have detrimental effects on brain function and structure provides a plausible explanation for the observed strong association between air pollution and an increased risk of cognitive defects, neurodevelopmental disorders, and neurodegenerative diseases [39]. Emerging evidence indicates that inflammation and oxidative stress are pivotal factors in the pathogenesis of disorders induced by air pollution. This is driven by the increased production of proinflammatory mediators and reactive oxygen species in response to exposure to various air pollutants [40].
In our study, the only significant causal association observed after applying a rigorous Bonferroni correction was between NO2 exposure and schizophrenia. Evidence from previous studies supports our finding. A comprehensive review mainly from Asian studies indicated increased risk was associated with short-term to PM2.5 (Risk ratio (RR) 1.0050, 95% CI 1.0017–1.0083), and NO2(RR 1.0275, 95% CI 1.0132–1.0420) [41]. A population-based cohort study in Denmark found that a 10-μg/m³ increase in childhood daily NO2 exposure and a 1-standard deviation increase in polygenic risk score were independently associated with an increased risk of schizophrenia [42]. Another study conducted in Denmark indicated that higher concentrations of residential NO2 and NOX exposure during childhood were associated with subsequently elevated risk of schizophrenia [43, 44]. A population-based cohort study from The Environmental-Risk Longitudinal Twin Study found that exposure to air pollution, particularly NO2 and NOx, was associated with increased odds of adolescent psychotic experiences [45]. Additionally, NO2 exposure may exacerbate the severity of schizophrenia. In Xi’an, a heavily polluted city in China, researchers found that short-term exposure to NO2 is associated with an increased risk of daily outpatient visits for schizophrenia [46]. Similar results were obtained in another cities in China [47, 48].
Long-term studies are susceptible to an increasing number of confounding factors, which can substantially weaken the final results of observational studies. Compared to observational studies, the MR method has advantages in studying time-varying exposures, especially long-term exposure, due to the principle that genetic variations are lifelong and constant. In this study, we introduced a novel GWAS-based method to investigate the causal relationship between air pollution and mental disorders. Our findings are not entirely consistent with previous research. Specifically, we did not find that air pollutants exerted significant adverse effects on depression. Additionally, the number of similar MR studies investigating the effects of air pollution on specific diseases has been increasing, including neurodegenerative diseases [49], cancers [50], and cardiovascular diseases [51]. With the increasing availability of open-access databases and summary-level data, more high-quality GWAS data will be better utilized to explore the effects of environmental substances on the human body.
While the larger sample size of the present study provided substantial statistical power, several limitations warrant consideration. First, the study focused on a select group of air pollutants and mental disorders, potentially limiting generalizability. Second, the predominantly European ancestry population restricts the applicability of findings to other ethnicities. Third, the limited sample overlap of participants between exposure and outcome data might raise concerns about selection bias. Fourth, in clinical practice, there is substantial overlap between mental disorders, such as bipolar disorder and anxiety disorder. However, pleiotropy among these disorders cannot be effectively identified or eliminated through conventional methods.
In conclusion, our findings support causal associations between exposure to ambient air pollution, particularly PM2.5, NO2, and NOx, and an increased risk of specific mental disorders. Given the selection criteria for instrumental variables and the adjusted p value results, these findings should be interpreted with caution. This still underscores the importance of developing effective mitigation strategies to reduce air pollution and its associated mental health and environmental burdens.
Data availability
All summary-level GWAS data used in this study were retrieved from open-access public databases for research purposes only. Summary-level GWAS data of air pollutants were downloaded from the IEU Open GWAS Project (https://gwas.mrcieu.ac.uk/), including PM2.5 (ukb-b-10817), PM2.5-10 (ukb-b-12963), PM10 (ukb-b-18469), NO2 (ukb-b-12417), and NOx (ukb-b-9942). Summary-level GWAS data of mental disorders were retrieved from the Psychiatric Genomics Consortium (https://pgc.unc.edu/).
References
Yin P, Brauer M, Cohen AJ, Wang H, Li J, Burnett RT, et al. The effect of air pollution on deaths, disease burden, and life expectancy across China and its provinces, 1990-2017: an analysis for the Global Burden of Disease Study 2017. Lancet Planet Health. 2020;4:e386–e98.
Lam HCY, Hajat S. Ambient temperature, air pollution and childhood bronchiolitis. Thorax. 2021;76:320–1.
Wang M, Zhou T, Song Y, Li X, Ma H, Hu Y, et al. Joint exposure to various ambient air pollutants and incident heart failure: a prospective analysis in UK Biobank. Eur Heart J. 2021;42:1582–91.
Brumberg HL, Karr CJ. Ambient air pollution: health hazards to children. Pediatrics. 2021;147;e2021051484.
Kim KH, Kabir E, Kabir S. A review on the human health impact of airborne particulate matter. Environ Int. 2015;74:136–43.
Verhoeven JI, Allach Y, Vaartjes ICH, Klijn CJM, de Leeuw FE. Ambient air pollution and the risk of ischaemic and haemorrhagic stroke. Lancet Planetary Health. 2021;5:e542–e52.
Anyanwu E. Complex interconvertibility of nitrogen oxides (NOX): impact on occupational and environmental health. Rev Environ Health. 1999;14:169–85.
Huang S, Li H, Wang M, Qian Y, Steenland K, Caudle WM, et al. Long-term exposure to nitrogen dioxide and mortality: a systematic review and meta-analysis. Sci Total Environ. 2021;776:145968.
Charlson F, van Ommeren M, Flaxman A, Cornett J, Whiteford H, Saxena S. New WHO prevalence estimates of mental disorders in conflict settings: a systematic review and meta-analysis. Lancet. 2019;394:240–8.
Hasin DS, Goodwin RD, Stinson FS, Grant BF. Epidemiology of major depressive disorder: results from the national epidemiologic survey on alcoholism and related conditions. Archiv Gen Psychiatry. 2005;62:1097–106.
Wittchen HU, Jacobi F, Rehm J, Gustavsson A, Svensson M, Jönsson B, et al. The size and burden of mental disorders and other disorders of the brain in Europe 2010. Eur Neuropsychopharmacol. 2011;21:655–79.
First MB. Diagnostic and statistical manual of mental disorders, 5th edition, and clinical utility. J Nervous Mental Dis. 2013;201:727–9.
Li D, Xie J, Wang L, Sun Y, Hu Y, Tian Y. Genetic susceptibility and lifestyle modify the association of long-term air pollution exposure on major depressive disorder: a prospective study in UK Biobank. BMC Med. 2023;21:67.
Nobile F, Forastiere A, Michelozzi P, Forastiere F, Stafoggia M. Long-term exposure to air pollution and incidence of mental disorders. A large longitudinal cohort study of adults within an urban area. Environ Int. 2023;181:108302.
Chen C, Liu C, Chen R, Wang W, Li W, Kan H, et al. Ambient air pollution and daily hospital admissions for mental disorders in Shanghai, China. Sci Total Environ. 2018;613-614:324–30.
Compa M, Baumbach C, Kaczmarek-Majer K, Buczyłowska D, Gradys GO, Skotak K, et al. Air pollution and attention in Polish schoolchildren with and without ADHD. Sci Total Environ. 2023;892:164759.
Yu X, Mostafijur Rahman M, Carter SA, Lin JC, Zhuang Z, Chow T, et al. Prenatal air pollution, maternal immune activation, and autism spectrum disorder. Environ Int. 2023;179:108148.
Sun Y, Headon KS, Jiao A, Slezak JM, Avila CC, Chiu VY, et al. Association of antepartum and postpartum air pollution exposure with postpartum depression in Southern California. JAMA Network Open. 2023;6:e2338315.
Sekula P, Del Greco MF, Pattaro C, Köttgen A. Mendelian randomization as an approach to assess causality using observational data. J Am Soc Nephrol. 2016;27:3253–65.
Ference BA, Holmes MV, Smith GD. Using Mendelian randomization to improve the design of randomized trials. Cold Spring Harbor Perspect Med. 2021;11:a040980.
Skrivankova VW, Richmond RC, Woolf BAR, Davies NM, Swanson SA, VanderWeele TJ, et al. Strengthening the reporting of observational studies in epidemiology using mendelian randomisation (STROBE-MR): explanation and elaboration. BMJ. 2021;375:n2233.
Sudlow C, Gallacher J, Allen N, Beral V, Burton P, Danesh J, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12:e1001779.
Howard DM, Adams MJ, Clarke TK, Hafferty JD, Gibson J, Shirali M, et al. Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nat Neurosci. 2019;22:343–52.
Otowa T, Hek K, Lee M, Byrne EM, Mirza SS, Nivard MG, et al. Meta-analysis of genome-wide association studies of anxiety disorders. Mol Psychiatry. 2016;21:1391–9.
Pantelis C, Papadimitriou GN, Papiol S, Parkhomenko E, Pato MT, Paunio T, et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–7.
Mullins N, Forstner AJ, O’Connell KS, Coombes B, Coleman JRI, Qiao Z, et al. Genome-wide association study of more than 40,000 bipolar disorder cases provides new insights into the underlying biology. Nat Genet. 2021;53:817–29.
Lee CH, Cook S, Lee JS, Han B. Comparison of two meta-analysis methods: inverse-variance-weighted average and weighted sum of Z-Scores. Genom Inform. 2016;14:173–80.
Kamat MA, Blackshaw JA, Young R, Surendran P, Burgess S, Danesh J, et al. PhenoScanner V2: an expanded tool for searching human genotype-phenotype associations. Bioinformatics. 2019;35:4851–3.
Giambartolomei C, Vukcevic D, Schadt EE, Franke L, Hingorani AD, Wallace C, et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet. 2014;10:e1004383.
Lin J, Zhou J, Xu Y. Potential drug targets for multiple sclerosis identified through Mendelian randomization analysis. Brain. 2023;146:3364–72.
Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50:693–8.
Wallace C. Eliciting priors and relaxing the single causal variant assumption in colocalisation analyses. PLoS Genet. 2020;16:e1008720.
Zuber V, Grinberg NF, Gill D, Manipur I, Slob EAW, Patel A, et al. Combining evidence from Mendelian randomization and colocalization: review and comparison of approaches. Am J Hum Genet. 2022;109:767–82.
Gibbs DC, Ward SV, Orlow I, Cadby G, Kanetsky PA, Luo L, et al. Functional melanoma-risk variant IRF4 rs12203592 associated with Breslow thickness: a pooled international study of primary melanomas. Br J Dermatol. 2017;177:e180–e2.
Peña-Chilet M, Blanquer-Maceiras M, Ibarrola-Villava M, Martinez-Cadenas C, Martin-Gonzalez M, Gomez-Fernandez C, et al. Genetic variants in PARP1 (rs3219090) and IRF4 (rs12203592) genes associated with melanoma susceptibility in a Spanish population. BMC Cancer. 2013;13:160.
Zaorska K, Zawierucha P, Nowicki M. Prediction of skin color, tanning and freckling from DNA in Polish population: linear regression, random forest and neural network approaches. Hum Genet. 2019;138:635–47.
Andersen JD, Johansen P, Harder S, Christoffersen SR, Delgado MC, Henriksen ST, et al. Genetic analyses of the human eye colours using a novel objective method for eye colour classification. Forensic Sci Int Genet. 2013;7:508–15.
Jacobs LC, Hamer MA, Gunn DA, Deelen J, Lall JS, van Heemst D, et al. A genome-wide association study identifies the skin color genes IRF4, MC1R, ASIP, and BNC2 influencing facial pigmented spots. J Investig Dermatol. 2015;135:1735–42.
Attademo L, Bernardini F. Air pollution as risk factor for mental disorders: in search for a possible link with Alzheimer’s Disease and Schizophrenia. J Alzheimer’s Dis. 2020;76:825–30.
Hahad O, Lelieveld J, Birklein F, Lieb K, Daiber A, Münzel T. Ambient air pollution increases the risk of cerebrovascular and neuropsychiatric disorders through induction of inflammation and oxidative stress. Int J Mol Sci. 2020;21:4306.
Song R, Liu L, Wei N, Li X, Liu J, Yuan J, et al. Short-term exposure to air pollution is an emerging but neglected risk factor for schizophrenia: a systematic review and meta-analysis. Sci Total Environ. 2023;854:158823.
Horsdal HT, Agerbo E, McGrath JJ, Vilhjálmsson BJ, Antonsen S, Closter AM, et al. Association of childhood exposure to nitrogen dioxide and polygenic risk score for Schizophrenia with the risk of developing Schizophrenia. JAMA Netw Open. 2019;2:e1914401.
Antonsen S, Mok PLH, Webb RT, Mortensen PB, McGrath JJ, Agerbo E, et al. Exposure to air pollution during childhood and risk of developing schizophrenia: a national cohort study. Lancet Planetary Health. 2020;4:e64–e73.
Qiu X, Danesh-Yazdi M, Wei Y, Di Q, Just A, Zanobetti A, et al. Associations of short-term exposure to air pollution and increased ambient temperature with psychiatric hospital admissions in older adults in the USA: a case-crossover study. Lancet Planetary Health. 2022;6:e331–e41.
Newbury JB, Arseneault L, Beevers S, Kitwiroon N, Roberts S, Pariante CM, et al. Association of air pollution exposure with psychotic experiences during adolescence. JAMA Psychiatry. 2019;76:614–23.
Liang Z, Xu C, Cao Y, Kan HD, Chen RJ, Yao CY, et al. The association between short-term ambient air pollution and daily outpatient visits for schizophrenia: a hospital-based study. Environ Pollut. 2019;244:102–8.
Bai L, Zhang X, Zhang Y, Cheng Q, Duan J, Gao J, et al. Ambient concentrations of NO(2) and hospital admissions for schizophrenia. Occup Environ Med. 2019;76:125–31.
Duan J, Cheng Q, Luo X, Bai L, Zhang H, Wang S, et al. Is the serious ambient air pollution associated with increased admissions for schizophrenia? Sci Total Environ. 2018;644:14–9.
Ning P, Guo X, Qu Q, Li R. Exploring the association between air pollution and Parkinson’s disease or Alzheimer’s disease: a Mendelian randomization study. Environ Sci Pollut Res Int. 2023;30:123939–47.
Li W, Wang W. Causal effects of exposure to ambient air pollution on cancer risk: Insights from genetic evidence. Sci Total Environ. 2024;912:168843.
Liang X, Liang L, Fan Y. Two-sample mendelian randomization analysis investigates ambient fine particulate matter’s impact on cardiovascular disease development. Sci Rep. 2023;13:20129.
Acknowledgements
The authors would like to thank Prof. Dongtao Lin of Sichuan University for copyediting this manuscript. We also wish to express our sincere gratitude to the databases that provide open GWAS data (IEU Open GWAS Project and Psychiatric Genomics Consortium). Your contributions are invaluable to the advancement of medical research.
Funding
This work was supported by the Ministry of Science and Technology of the People’s Republic of China (No. 2022ZD0211700), the 135 Project from West China Hospital of Sichuan University (Nos. 2023HXFH006, 2023HXFH040), China Postdoctoral Science Foundation (No. GZC20231802) and Sichuan University (No. 2022SCUH0023).
Author information
Authors and Affiliations
Contributions
Huanhuan Fan: Conceptualization, Software, Writing - Original Draft. Junhong Li: Methodology, Software, Data Curation, Writing - Original Draft. Yikai Dou: Data Curation, Software, Funding acquisition. Yushun Yan: Methodology. Min Wang: Validation. Xiao Yang: Conceptualization, Validation, Writing - Review & Editing, Funding acquisition. Xiaohong Ma: Conceptualization, Writing - Review & Editing, Funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All original studies had obtained ethical approval. This study used publicly available summary data from the original studies, which obviated the need for ethical approval.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fan, H., Li, J., Dou, Y. et al. Linking ambient air pollution to mental health: evidence based on the two-sample Mendelian randomization and colocalization study. Transl Psychiatry 14, 489 (2024). https://doi.org/10.1038/s41398-024-03196-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-024-03196-0