Abstract
Spinal cord injury (SCI) remains a significant therapeutic challenge, lacking effective treatment options. Related studies have found that neonatal microglia are more effective than adult microglia in promoting the recovery of SCI, but the reason why neonatal, not adult, microglia are more conducive to SCI recovery is not clear, the differences of gene expression and pathways between them are still worth exploring. Therefore, we examined changes in the microglial transcriptome after SCI in neonatal and adult mice. We identified hub genes or pathways that exhibited significant differential expression between the two groups. Four Gene sets were established for further analysis, named Gene set 1, Gene set 2, Gene set 3, Gene set 4, respectively. GO analysis revealed enrichment in categories critical for injury repair, including DNA metabolism, replication, recombination, meiotic cell cycle progression, regulation of cell-cell adhesion, megakaryocyte and endothelial development, modulation of the neuroinflammatory response, endocytosis, and regulation of cytokine production and cell migration. KEGG analysis revealed enrichment in pathways critical for various cellular processes, including the p53, TNF, PI3K-AKT, PPAR and B cell receptor signaling pathway, axon guidance, cytokine-cytokine receptor interaction. PPI and TF-hub gene-microRNA networks were constructed to elucidate the underlying gene regulatory mechanisms. Additionally, drug prediction was performed to identify potential therapeutic candidates. Finally, 11 hub genes (Chek1, RRM2, Lyve1, Mboat1, Clec4a3, Ccnd1, Cdk6, Zeb1, Igf1, Pparg, and Cd163) were selected from four Gene sets for further validation using qRT-PCR. We identified candidate genes and pathways involved in microglial transcriptome heterogeneity after SCI in neonatal and adult mice. These findings provide valuable insights into potential therapeutic targets for neonatal microglia in the treatment of SCI.
Similar content being viewed by others
Introduction
Spinal cord injury (SCI), a devastating disease of the central nervous system (CNS), is characterized by severe functional loss that leads to long-term disability and associated complications1,2. The complex pathophysiology after SCI significantly impedes functional recovery3. Current treatment strategies, including acute surgical decompression, pharmacological interventions, and cell therapy coupled with neurorehabilitation, prevent secondary injury and decelerate disease progression4,5. However, these approaches do not promote neurological regeneration, resulting in a lack of curative therapies for SCI6,7,8.
Recent studies have revealed significant restoration of neural and motor function in neonatal mice after SCI. Notably, there are obvious differences in the recovery of neonatal mice and adult mice after SCI, which may be related to the differences in the expression of genes and related substances between them. The expression of neurotransmitters in glutamatergic interneurons in neonates is excitatory phenotype, but in adults it is inhibitory. Related single-cell sequencing has also found that some cells with ependymal gene expression profiles are only significantly enriched in neonatal injured spinal cord9,10. SCI in neonates exhibits significant upregulation of genes associated with axon growth, cell proliferation, and myelination, this spontaneous functional recovery observed in neonates is reduced in adults, which may be attributed to the expression of genes in most adult neurons that restrict axon growth and limit the energy available for regeneration11,12,13. In addition to neurons, glial cells also play an important role in the success or failure to SCI recovery in neonatal or adult mice, respectively14. Further studies are necessary to elucidate the contribution of reactive glial cells to the different regenerative capacity observed between neonatal and adult SCI.
Microglia, resident macrophages in the CNS, play a pivotal role in SCI recovery, particularly in neonates15,16,17. The results of one study have suggested that microglia in neonatal mice play an important role in coordinating SCI recovery. Transient activation of neonatal microglia promotes scar-free healing, and transplantation of neonatal microglia can significantly improve spinal cord lesions in adults18. In adult mice, although many studies have affirmed the therapeutic effect of microglia on SCI, the recovery of SCI in adults cannot achieve significant results due to the differentiation of microglia into the classically activated microglia (M1) and its pro-inflammatory effect19. Therefore, it may be worth studying the differences in microglial transcriptome genes between neonatal mice and adult mice after SCI. This approach will provide valuable insights into the differential microglial response between neonates and adults, revealing novel therapeutic targets for SCI.
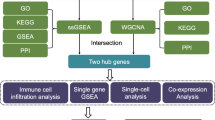
We used two publicly available high-throughput sequencing microglial datasets from the Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/geo/)20. These datasets included GSE150871 (neonatal mice) and GSE113566 (adult mice). Differentially expressed genes (DEGs) were identified via selection of samples at the corresponding 3 days post-injury (dpi) time point within each dataset. Subsequently, DEGs that exhibited discordant expression patterns between the two groups were isolated. Functional enrichment analysis of the screened DEGs was performed using Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways to elucidate relevant biological functions and enriched pathways. Next, we constructed a protein-protein interaction (PPI) network using Cytoscape software to identify hub genes. Furthermore, we constructed a transcription factor (TF)-hub gene-microRNA network to explore the regulatory mechanisms underlying these hub genes. A drug profile database was used to identify potential therapeutic drugs and compounds that target the identified hub genes. Finally, quantitative real-time polymerase chain reaction (qRT-PCR) was used to validate the expression levels of hub genes in neonatal and adult mouse SCI models. Figure 1 shows the schematics of the experimental procedures and validation strategies.
Materials and methods
Mice
In this experiment, animals were 7-week-old female C57BL/6 mice and 2-day-old female C57BL/6 mice (Jinan Pengyue Biotechnology Co., Ltd., Jinan, China). Mice were housed under controlled conditions, including a 12-hour light/dark cycle, a temperature of 23 ± 2 °C, a relative humidity of 50 ± 5%, and free access to food and water at all times. All animal procedures, including euthanasia, were performed in accordance with the animal care regulations of Qingdao University. All animal procedures were conducted in accordance with the protocol approved by the Laboratory Animal Welfare Ethics Committee of Qingdao University (NO.202403C572020240620116)and the guidelines for the use of animals in the study according to the Animal Research: In Vivo Experiment Report (ARRIVE). All surgeries are performed under anesthesia and every effort is made to reduce the pain of the animal.
Data acquisition
Microglial transcriptomic data were obtained from the GEO database, including two datasets (GSE150871 and GSE113566) generated using the GPL17021 (Illumina HiSeq 2500, Mus musculus) platform. GSE150871 comprises 5 samples from neonatal mouse microglia (postnatal day 2) at three time points post-injury (0, 3, and 5 dpi). We selected samples from 0 (control) and 3 dpi for further analysis. Dataset GSE113566 investigated transcriptional changes in microglia of adult mice (6–8 weeks old) after SCI at 3, 7, and 14 dpi. Samples from 0 (control) and 3 dpi were also selected for subsequent analyses.
DEG analysis
Microglial gene expression profiles between 3 dpi and control samples in GSE150871 were compared using the Seurat (version 5.0.1, https://github.com/satijalab/seurat) and harmony packages within R statistical software (version 4.3.2)21. This analysis yielded an expression matrix of microglia-related genes, followed by DEG analysis. DEGs were defined as genes with a | log2(fold change) | > 1 and an adjusted p-value < 0.05. For GSE113566, the DESeq2 package in R was used to identify DEGs between 3 dpi and control samples. DEGs were defined using similar criteria (| log2(fold change) |> 1 and adjusted p-value < 0.05). Volcano plots for DEGs were generated using the online Chiplot platform (https://www.chiplot.online/Volcano_plot.html)22. Heatmaps depicting DEG expression patterns were constructed using the ComplexHeatmap package (version 2.13.1)23.
Comparison of DEGs between GSE150871 and GSE113566
To examine changes in gene expression profiles between the two datasets, we performed Venn diagram analysis using a bioinformatics platform designed for data analysis and visualization (https://www.bioinformatics.com.cn/en?p=5)24. We calculated the intersection of upregulated and downregulated DEGs in both datasets. Subsequently, these screened genes (named Gene set 1–4) were subjected to GO and KEGG pathway enrichment analyses.
Functional enrichment analysis of identified gene sets
GO enrichment analysis categorized genes based on their biological processes (BPs), cellular components (CCs), and molecular functions (MFs). KEGG enrichment analysis focused on identifying enriched pathways within the dataset. The Metascape database (https://metascape.org/) was used for GO and KEGG enrichment analyses25. Stringent criteria were applied, with a minimum overlap of 3 genes and a minimum enrichment score of 1.5. Pathways with p-values < 0.01 were considered significantly enriched. Metascape automatically clustered the enriched terms, and the term with the highest ˗ log(p-value) within each category was selected as the representative. The top 20 enriched GO terms and KEGG pathways were visualized using a combination of bubble diagrams, string diagrams, and Sankey diagrams generated with a bioinformatics platform (https://www.bioinformatics.com.cn/en).
PPI network
To identify interacting genes and potential hub genes, we constructed a PPI network using the Search Tool for the Retrieval of Interacting Genes (STRING, https://cn.string-db.org/) database26. Interactions with a combined score > 0.4 were considered significant. The resulting PPI network data were retrieved and visualized using Cytoscape software (version 3.8.0)27. Three centrality measures, including betweenness, closeness, and degree, were used for hub gene selection. Hub genes were selected based on the cumulative score obtained from all three centrality measures. The top 10–20 genes with the highest total scores were identified as hub genes through the Cytohubba plugin (http://apps.cytoscape.org/apps/cytohubba) in Cytoscape software.
Construction of TFs-hub Gene-miRNAs network and identification of drugs and compounds
We used the miRwalk database (http://miRwalk.umm.uni-heidelberg.de/) to identify potential miRNAs that regulate hub genes28. The analysis was restricted to miRNAs predicted to target the 3’ untranslated region of hub genes with a miRwalk score of ≥ 0.95. To ensure accuracy, miRNA interactions were validated using the miRTarBase database. The NetworkAnalyst 3.0 platform (https://www.networkanalyst.ca/) was used to predict TFs regulating hub genes29. TF-gene interactions were extracted from ENCODE ChIP-seq data using the BETA Minus algorithm. Only TF-gene interactions with a peak intensity signal < 500 and predicted regulatory potential score < 1 were used30,31. Finally, we constructed and visualized the TF-hub gene-miRNA regulatory network using Cytoscape software.
The Enrichment platform (https://maayanlab.cloud/Enrichr), linked to the Drug Signatures database, was used to predict drugs and compounds targeting the hub genes32,33. The top 10 results were ranked by their p-values and visualized as a bar chart.
SCI model
Female C57BL/6 mice aged 7 weeks and female neonatal C57BL/6 mice aged 2 days were used to establish SCI models17,34. After an intraperitoneal injection of 80 mg/kg pentobarbital for anesthesia, the surgical area was shaved and disinfected with 75% ethanol; a midline incision was made in the thoracic vertebra. Laminectomy was performed at the T10 vertebral level to expose the spinal cord. Tips of 0.1-mm forceps were carefully inserted on either side of the spinal cord to include the full width of the spinal cord. Then, the spinal cord was fully crushed for 2 s, while the dura matter was kept intact. The muscles and skin layers were sutured, and the mice were placed on a heating pad until full recovery. Manual bladder emptying was performed twice daily.
RNA extraction and qRT-PCR analysis
Spinal cord tissues were harvested from mice at 3 days post-SCI for RNA analysis. Mice were anesthetized with an overdose of pentobarbital intraperitoneally and then perfused with pre-chilled PBS through the heart until the mice’s liver and effluent blood become transparent, mice died painlessly. Quickly dissect a 1 cm piece of spinal cord (centered on the T10 laminectomy site) and place in liquid nitrogen for later use. Total RNA was extracted using TRIzol® reagent (Invitrogen, Carlsbad, CA, USA). Reverse transcription of RNA to complementary DNA (cDNA) was performed using the All-in-One First-Strand Synthesis Master Mix Kit (share-Bio, Shanghai, China). Next, qRT-PCR was performed using ChamQ Universal SYBR qPCR Master Mix (Vazyme, Nanjing, China). Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control for the normalization of expression levels, and the 2-ΔΔCT method was used to determine the relative expression levels of target genes. Primer sequences used for qRT-PCR are presented in Table 1.
Statistical analysis
GraphPad Prism software (version 8.0.2; GraphPad Software, San Diego, CA, USA) was used for statistical analysis of qRT-PCR data. Student’s t-test was used to compare gene expression levels between groups. Data are presented as means ± standard errors of the mean. P-values < 0.05 were considered statistically significant.
Results
Identification of DEGs after SCI
Two publicly available transcriptomic datasets (GSE113566 and GSE150871) were retrieved from the GEO database to identify DEGs after SCI in neonatal and adult mice. For GSE113566, transcriptome data from mice (6–8 weeks old) at 3 days post-SCI were analyzed. In total, 1,281 DEGs, including 856 upregulated and 425 downregulated genes, were identified with an adjusted p-value < 0.05 and | log2(fold change) | > 1 (Fig. 2A). Similarly, for GSE150871, transcriptome data from neonatal mice (postnatal day 2) at 3 days post-SCI were analyzed using R software packages. In total, 1,458 DEGs, including 1,010 upregulated and 448 downregulated genes, were identified with adjusted p-value < 0.05 and | log2(fold change) | > 1 (Fig. 2B). Heatmaps were generated to visualize the expression patterns of DEGs in each dataset (Fig. 2C and D).
Analysis of DEGs after SCI in GSE113566 and GSE150871 datasets. (A) Volcano plot depicting the distribution of 1,281 DEGs in the GSE113566 dataset. Pink points represent upregulated genes, whereas green points indicate downregulated genes. (B) Volcano plot showing the distribution of 1,458 DEGs in the GSE150871 dataset. Yellow points represent upregulated genes, whereas blue points indicate downregulated genes. (C) Heatmap showing the distribution of DEGs in the GSE113566 dataset. The color gradient from pink to green indicates the relative expression level, with pink and green indicating upregulated and downregulated expression, respectively. (D) Heatmap illustrating the distribution of DEGs in the GSE150871 dataset. The color gradient from yellow to blue represents the relative expression levels, with yellow and blue indicating upregulated and downregulated expression, respectively.
Comparative analysis of DEGs after SCI between neonatal and adult mice
To examine the DEG changes after SCI between neonatal and adult mice, we used a Venn diagram to visualize the overlap between DEGs identified in datasets GSE113566 and GSE150871. We compared upregulated DEGs in neonates with upregulated and downregulated DEGs in adults. This analysis revealed 196 DEGs upregulated in both groups, 11 DEGs upregulated in neonates but downregulated in adults (Gene set 1), and 803 DEGs upregulated exclusively in neonates but not differentially expressed in adults (Gene set 3) (Fig. 3A, C, and E). Similarly, we compared downregulated DEGs in neonates with those in adults. We identified 48 DEGs downregulated in both groups, 38 DEGs downregulated in neonates but upregulated in adults (Gene set 2), and 362 DEGs downregulated exclusively in neonates but not differentially expressed in adults (Gene set 4) (Fig. 3B, D, and F). Consequently, the above four Gene sets we considered worthy of further analysis.
Venn diagram depicting the overlap of DEGs identified in neonatal and adult mice. Gene set 1 (11 DEGs) is upregulated in neonates but downregulated in adult mice. Gene set 2 (38 DEGs) is downregulated in neonates but upregulated in adult mice. Gene set 3 (803 DEGs) is upregulated in neonates but not differentially expressed in adult mice. Gene set 4 (362 DEGs) is downregulated in neonates but not differentially expressed in adult mice.
Functional enrichment analysis of gene sets
To elucidate the potential biological functions and underlying mechanisms associated with the four identified Gene sets, we used GO and KEGG enrichment analyses. Due to the limited number of DEGs in Gene set 1, GO or KEGG analyses were not applicable. Therefore, we summarized the specific information and reported functions of these genes based on a comprehensive literature review (Table 2). For Gene set 2, GO enrichment analysis revealed significant enrichment of BPs related to DNA metabolism, replication, mitotic cell cycle progression, regulation of cell cycle phase transitions, homologous recombination, and base-excision/postreplication repair (Fig. 4A–B; Table 3). Gene set 3, including 803 DEGs, exhibited significant enrichment of BPs associated with immune regulation and response (cell activation, leukocyte migration), interleukin (IL)-1 and tumor necrosis factor (TNF) superfamily cytokine production, mitogen-activated protein kinase cascade signaling, apoptosis, neuroinflammation, cell-cell adhesion, and endocytosis (Fig. 4C–D; Table 3). Gene set 4 (362 DEGs) exhibited enrichment of BPs involved in DNA metabolism, double-strand break repair via homologous recombination, cell cycle regulation, cell morphogenesis, and cell adhesion (Figs. 4E–F; Table 3). Detailed enrichment results for molecular function and cellular component categories are provided in Supplementary file. KEGG pathway analysis of Gene set 2 (38 DEGs) identified significant enrichment of pathways associated with DNA replication, homologous recombination, cell cycle, base excision repair, and p53 signaling (Fig. 5A; Table 4). Furthermore, Gene set 3 exhibited enrichment of key immune signaling pathways, including TNF signaling, B cell receptor signaling, phosphoinositide 3-kinase/protein kinase B (PI3K-AKT) signaling, and peroxisome proliferator-activated receptors (PPAR) signaling pathways (Fig. 5B; Table 4). The main enrichment pathways in Gene set 4 included those involved in cell cycle regulation (Cell cycle pathway), DNA repair (Fanconi anemia pathway), DNA replication, microRNA regulation in cancer, adipocyte lipolysis regulation, and axon guidance (Fig. 5C; Table 4).
GO enrichment analysis. (A,C,E) Bubble diagrams depict GO term enrichment analysis. Horizontal and vertical axes represent fold enrichment and specific GO pathways, respectively. Bubble size corresponds to the number of enriched genes. The color gradient transitions from red to blue, denoting enrichment significance (high to low). (B,D,F) String diagrams depict the distribution of screened DEGs across various enriched GO functions. The left side of each plot indicates the DEGs, whereas the right side represents the different GO terms. Connecting bands visualize genes assigned to their corresponding GO terms. (A,B) Major enriched BPs associated with 38 DEGs in Gene set 2. (C,D) Major enriched BPs associated with 803 DEGs in Gene set 3. (E,F) Major enriched BPs associated with 362 DEGs in Gene set 4.
KEGG pathway enrichment analysis. Bubble diagrams depict enriched KEGG pathways, KEGG pathway Image usage copyright was obtained through Kanehisa laboratories102,103,104,105. Horizontal and vertical axes represent the gene ratio and specific KEGG pathway names, respectively. Bubble size corresponds to the number of enriched genes. The color gradient transitions from red to blue indicate enrichment significance (high to low). Sankey diagrams illustrate the distribution of screened DEGs across various enriched KEGG pathways. The left side of the plot represents the DEGs, whereas the right side indicates the different KEGG terms. Connecting bands visualize genes assigned to their corresponding KEGG terms. (A) KEGG pathways associated with 38 DEGs in Gene set 2 (total of 5 pathways). (B) KEGG pathways associated with 803 DEGs in Gene set 3 (total of 20 pathways). (C) KEGG pathways associated with 362 DEGs in Gene set 4 (total of 6 pathways).
PPI network analysis and hub gene identification
To elucidate the complex regulatory mechanisms underlying SCI, we used PPI network analysis to reveal functional relationships, characteristics, and biological pathways between DEGs. We constructed PPI networks in the STRING database using 38, 803, and 362 DEGs from Gene sets 2, 3, and 4, respectively. The PPI network for Gene set 2 (38 nodes, 326 edges) exhibited a high average local clustering coefficient (0.727) and significant PPI enrichment (p-value < 1.0e-16). Similarly, the PPI networks for Gene sets 3 (730 nodes, 3014 edges) and 4 (327 nodes, 397 edges) displayed high average local clustering coefficients (0.412 and 0.325, respectively) and significant PPI enrichment (p-value < 1.0e-16). These networks were visualized in Cytoscape (Fig. 6A, C, and E). Node size and color intensity were correlated with degree (number of interacting partners); larger, darker nodes represented higher-degree proteins. Subsequently, Cytoscape plugins using degree, closeness, and betweenness algorithms were used to identify hub genes within each network (Fig. 6B, D, and F). The top 10–20 genes with the highest total scores from these algorithms were designated as hub genes (Table 5). Finally, we compiled a list of 61 differentially expressed hub genes, including 50 from Gene sets 2–4 and 11 from Gene set 1 (Table 6). These 61 genes are considered key microglia-dependent genes with the most important roles in SCI repair.
PPI network construction and differentially expressed hub gene identification. (A,C,E) PPI networks constructed using the cytoNCA plugin. The size and color depth of each node correspond to its “degree” value (number of connections). (B,D,F) PPI networks constructed using the cytoHubba plugin, with three algorithms of betweenness, closeness, and degree. The color gradient from red to yellow indicates the scores assigned by each algorithm (high to low).
Construction of TF-hub gene-miRNA network and drug prediction
To further investigate the potential regulatory mechanisms associated with SCI, we analyzed the interactions between TFs and miRNAs of the 61 differentially expressed hub genes. We uploaded these genes to the NetworkAnalyst platform to predict and establish a TF-hub gene network. In total, 43 hub genes interacted with TFs, forming a network with 76 nodes and 220 interactions (Fig. 7A). Next, we used miRwalk to construct a hub gene-miRNA network encompassing 53 nodes and 83 interactions (Fig. 7B). Subsequently, both networks were integrated into Cytoscape to generate a comprehensive TF-hub gene-miRNA network diagram (Fig. 7C). The construction of TF-hub-miRNA network diagram reveals the complex interaction between transcription factors and microRNAs in gene expression regulation, which is helpful for understanding the molecular mechanism of gene expression regulation in specific disease states. Transcription factors regulate the transcription rate of genes by binding to specific DNA sequences, while miRNAs regulate gene expression at the post-transcriptional level by degrading target gene mRNA or inhibiting its translation, and they are involved in the regulation of driving disease processes and may therefore be potential drug targets.
TF-hub gene-miRNA network, drug prediction and qRT-PCR validation of hub genes. (A) TF-hub gene network (red V-shapes: hub genes, purple ellipses: TFs). (B) Hub gene- miRNA network (red ellipses: hub genes, green ellipses: miRNAs). (C) Integrated TF-hub gene-miRNA network (red V-shapes: hub genes, purple ellipses: TFs, green triangles: miRNAs). (D) The top 10 candidate drugs identified by 61 hub genes. The color gradient from brighter to darker indicates the significance of each drug (high to low). (E) Genes from Gene sets 1–4 subjected to further validation using qRT-PCR. (F) qRT-PCR validation of 11 genes (from panel (E)) in neonatal mice after SCI. (G) qRT-PCR validation of 11 genes (from panel (E)) in adult mice after SCI. Data are expressed as means ± standard errors of the mean (SEMs) (n = 3). *P < 0.05, **P < 0.01, and ***P < 0.001.
Furthermore, we investigated potential therapeutic interventions by identifying drugs and compounds that target these microglia-dependent DEGs. The top 10 candidate drugs included troglitazone, resveratrol, bisphenol A, indole-3-carbinol, PD98059, genistein, dasatinib, dexamethasone, phorbol-12-myristate-13-acetate, and methaneselenic acid (Fig. 7D), then we listed the details of the genes targeted by each drug(Table 7).
qRT-PCR validation of hub genes
To validate the identified hub genes and assess their biological relevance, we performed qRT-PCR on a selection of 11 genes from the 61 microglia-dependent DEGs. These genes (Chek1, Rrm2, Lyve1, Mboat1, Clec4a3, Ccnd1, Cdk6, Zeb1, Igf1, Pparg, and Cd163) were selected by reviewing the comprehensive literature and considering their potential therapeutic role in SCI, with 2–3 genes from each Gene set (Fig. 7E)35,36,37,38,39,40,41,42. These genes have been implicated in promoting SCI recovery in mouse models. qRT-PCR analysis largely confirmed the expression patterns observed in the sequencing data. For example, Chek1 and RRM2 exhibited downregulation in neonatal mice but upregulation in adult mice after SCI. Clec4a3 expression was upregulated in neonates but downregulated in adults. Also consistent with the analysis results, Ccnd1 and Zeb1 were downregulated in neonatal mice and not differential expressed in adults after SCI (Fig. 7F-G).
Discussion
Despite advancements in modern medicine, a definitive cure for SCI remains elusive. This situation presents a significant challenge to global healthcare systems, primarily due to the currently limited understanding of cellular responses to SCI43. Neonatal mice exhibit remarkable regenerative capacity after SCI, and transplantation of microglia isolated from neonates promotes regeneration in adult mice. However, the mechanisms underlying this repair-promoting effect of neonatal microglia remain unclear18,44. Identification of transcriptomic differences between microglia from neonates and adults after SCI could reveal novel therapeutic targets for microglia-dependent treatment strategies. We utilized bioinformatics analysis to compare microglial transcriptomic changes in neonatal and adult mice at 3 days post-SCI. This analysis revealed DEGs, which were subsequently validated through in vivo experiments.
We compared DEGs in neonatal mice with DEGs in adult mice after SCI. Four Gene sets were selected for further analysis, including genes upregulated in neonates but downregulated in adults, genes downregulated in neonates but upregulated in adults, genes upregulated in neonates but not differentially expressed in adults, and genes downregulated in neonates but not differentially expressed in adults. GO and KEGG enrichment analyses revealed enrichment terms in these Gene sets primarily associated with DNA transcription and replication, cell differentiation and regeneration, post-injury immune response, and neuroinflammation.
Acute SCI elicits a robust immune response and neuroinflammation, key contributors to secondary injury. These processes begin within hours of the initial insult and persist for months within the lesion site45. Ischemia, oxidative stress, edema, and excitotoxic glutamate release initiate the cascade within a few hours after injury. Subsequently, a diverse array of inflammatory cells infiltrates the peri-lesional area, triggering substantial upregulation of pro-inflammatory cytokines, including IL-1β, TNF-α, and IL-646,47. At the same time, limited spontaneous repair processes initiate in neonatal and adult mice after primary or secondary injury-induced tissue and cell necrosis48. Consistent with these observations, our enrichment results revealed significant enrichment of DEGs in our identified Gene sets. These DEGs were functionally associated with DNA replication, homologous recombination, regulation of mitotic cell cycle phase transition, cell morphogenesis, base excision repair, cell proliferation and apoptosis, immune response regulation, leukocyte migration and activation, regulation of IL-1 and TNF production, and modulation of neuroinflammatory responses. Notably, several signaling pathways, such as p53, PI3K-AKT, PPAR, and AMP-activated protein kinase (AMPK) pathways, were highly associated with SCI repair through their roles in the regulation of immune inflammation, cell proliferation, apoptosis, and regeneration.
The p53 protein regulates diverse biological processes depending on the nature of cellular stress signals. Recent studies have characterized its involvement in numerous biological pathways, including autophagy, cell metabolism, ferroptosis, and reactive oxygen species production49. Moreover, p53 regulation can promote SCI recovery by regulating antioxidant defenses, apoptosis, and ferroptosis50,51. Ferroptosis, an iron-dependent form of programmed cell death, contributes to organ dysfunction and degeneration52,53. Increased free iron levels exacerbate SCI by inducing glutamate excitotoxicity in spinal motor neurons. Conversely, iron chelation demonstrates therapeutic potential in alleviating SCI54. Recent studies have implicated p53 in ferroptosis regulation. Notably, inhibition of dihydroorotate dehydrogenase (DHODH) can protect spinal cord neurons from ferroptosis by modulating p53 activity, thereby suppressing arachidonic acid 15-lipoxygenase (ALOX15), and promoting neuronal survival and SCI recovery52. Additionally, two other signaling pathways, PI3K/AKT and PPAR-γ, are significantly upregulated in neonatal mice and contribute to various cellular functions, including cell proliferation, survival, and inflammation55,56. Activation of the PI3K/AKT/mTOR pathway with resveratrol has demonstrated significant improvement in neurological function through reductions of cerebral infarction volume and neuronal damage57. PPAR-γ, a ligand-activated nuclear receptor, regulates glucose and lipid metabolism, endothelial function, and inflammation58,59,60. PPAR agonists have demonstrated significant effectiveness in reducing neuronal loss and improving motor function recovery after SCI61,62,63,64. The AMPK pathway, a key regulator of cellular metabolism and inflammation, plays a crucial role in SCI. AMPK regulates NLRP3 inflammasome assembly and activation, influencing inflammation and oxidative stress in various diseases65,66. Modulation of the AMPK signaling pathway can reduce oxidative stress, regulate spinal cord and neuronal glucose metabolism, and promote functional recovery post-SCI67. Therefore, genes associated with these pathways may represent promising therapeutic targets for SCI. For example, CCND1 and Cdk6, genes involved in p53 and PI3K/AKT pathways, warrant further studies. CCND1, a critical cyclin protein, exhibits increased expression 24 h post-injury. It activates CDK4/6, triggering a cascade of events that promote neuronal apoptosis and glial cell reactive proliferation, thereby contributing to the secondary injury process in SCI6,68. Notably, the CDK inhibitor flavopiridol demonstrates potential in restoring cell cycle control. Delayed administration of this drug significantly reduces the lesion volume, preserves white matter integrity, improves motor function recovery after SCI, and mitigates secondary tissue damage69,70.
TFs and miRNAs can regulate gene expression at the transcriptional level. Our TF analysis identified Myc as a potential key player in SCI. c-Myc, a member of the Myc oncogene family, critically regulates cell proliferation and differentiation. Several studies of traumatic SCI have demonstrated that c-Myc knockdown reduces oligodendrocyte precursor cell proliferation and promotes their differentiation and myelination capacity in vitro71. Furthermore, our network analysis revealed several miRNAs with important regulatory roles in SCI. For example, miR-329-3p targets Pik3r1, a gene downregulated in neonatal mice after SCI. Several studies have revealed that miR-329-3p inhibition can reverse lipopolysaccharide-induced neuronal apoptosis, inflammation, and oxidative stress. Conversely, miR-329-3p overexpression attenuates the neuroprotective effects of mesenchymal stem cell-derived exosomes in injured spinal cords72. The upregulated DEG Kit in neonatal mice interacts with miR-222-3p. Notably, miR-222-3p upregulation can inhibit neuronal apoptosis and promote motor function recovery after SCI73. Furthermore, miR-124-3p, the most enriched miRNA in neuron-derived exosomes, interacts with three hub genes: Anxa5, Cdk6, and Chek1. In vivo and in vitro studies have demonstrated that miR-124-3p suppresses M1 microglia and A1 astrocyte activation by inhibiting myosin heavy chain 9 (MYH9) activity, significantly promoting functional recovery after SCI74. Additionally, miR-124-3p promotes M2 macrophage polarization, reducing tissue injury in spinal cord ischemia-reperfusion injury and alleviating neurogenic pain in SCI mice75,76. These findings represent only a fraction of the miRNAs and TFs identified in the SCI repair network. Further studies are needed to explore novel therapeutic targets in SCI.
Resveratrol, a natural polyphenol with antioxidant, anti-inflammatory, antibacterial, and anti-neurodegenerative properties, has emerged as a potential therapeutic candidate; it can promote functional recovery after SCI by modulating gut microbiota and inhibiting microglial activation77,78,79,80,81. Extracellular signal-regulated kinase (ERK) inhibition plays a crucial role in the neuroprotective effects of various drugs82. PD-98,059, a predicted ERK inhibitor, could potentially alleviate neuropathic pain by enhancing ERK inhibition83. Genistein, a natural soy-derived phytoestrogen with anti-inflammatory, antioxidant, and neuroprotective effects, may reduce lipid peroxidation, oxidative damage, and inflammatory response after SCI84,85,86,87. Dexamethasone, a commonly used glucocorticoid, has a well-established role in SCI treatment. However, the efficacies of troglitazone, bisphenol A, indole-3-carbinol, dasatinib, and methane-selenic acid require further investigation.
We identified 11 genes potentially influencing SCI recovery from a pool of 61 DEGs for further verification using qRT-PCR. These genes were Chek1, RRM2, Lyve1, Mboat1, Clec4a3, Ccnd1, Cdk6, Zeb1, Igf1, Pparg, and Cd163. The expression profiles of Chek1, RRM2, Ccnd1, Mboat1, Clec4a3, Igf1, and Cd163 were consistent with the RNA sequencing results in neonatal mice after SCI. Notably, three genes (Chek1, RRM2, and Clec4a3) exhibited expression trends similar to those observed in adult mice. Chek1 is a well-established regulator of cell proliferation and apoptosis, the upregulation of Chek1 may indicate cellular senescence and coordinate DNA damage88,89. However, its specific contribution to SCI remains unclear. RRM2 plays a multifaceted role in regulating purine metabolism, the cell cycle, and DNA repair. The results of recent studies suggest its involvement in protein regulation and modification, as well as angiogenesis90. Notably, RRM2 exhibits a specific elevation in liver cancer and functions as an endogenous ferroptosis suppressor by stimulating glutathione synthesis via glutathione synthase91. This indicates the potential of serum RRM2 as a biomarker for ferroptosis diagnosis and treatment. Considering the emerging role of ferroptosis in various diseases, including SCI, RRM2 warrants further investigation as a potential therapeutic target for SCI. Clec4a3 is a C-type lectin gene within the Aplec cluster. One study has demonstrated that Aplec, which is widely expressed in microglia, upregulated under inflammatory stimuli, is involved in microglial signaling after SCI, however, its expression has no effect on glial cell activation. Moreover, it regulates motor neuron survival and lymphocyte infiltration, indicating its crucial role in modulating immune responses and neuronal cell survival92. Furthermore, Ccnd1, Mboat1, Igf1, and Cd163 emerged as potential therapeutic targets, particularly in neonatal mice. Cyclin D1 (Ccnd1), is upregulated in neurons and microglia after traumatic brain injury (TBI), and its downregulation has important implications for the improvement of neurodegeneration, the recovery of cognitive function, the reduction of lesion volume, and the activation of cortical microglia after TBI93. Mboat1, a phospholipid-modifying enzyme, is particularly intriguing. Ferroptosis, a form of cell death mediated by iron-dependent phospholipid peroxidation and plasma membrane rupture, has been implicated in SCI. Mboat1/2 function as ferroptosis inhibitors by remodeling phospholipids94,95. This suggests a potential neuroprotective role for Mboat1 in SCI, highlighting the need for further studies. Igf1 is a protein hormone with neurotrophic properties. It plays a critical role in CNS development, promoting neuronal differentiation and survival during normal development and aiding recovery after injury or disease96. Numerous studies have revealed its pleiotropic benefits in the CNS, including the promotion of SCI repair by inducing a shift in microglial polarization towards neuroprotective and anti-inflammatory phenotypes97,98. In the chronic stages of SCI, Igf1 therapy may reverse permanent hippocampal alterations and improve cognitive deficits99. It has also been shown to be significantly upregulated in microglia subclusters that are upregulated in ischemic brain tissue, and its high expression can transform microglia into a neuroprotective phenotype100. Similarly, the difference in the expression of this gene in microglia between neonatal mice and adult mice after SCI may be one of the reasons why neonatal mice recover better after injury than adult mice.
Our study has several limitations. First, considering the relatively small animal sample size, hub gene expression should be confirmed in a larger cohort. Second, we solely focused on data from the third day post-SCI. Examination of DEGs across multiple time points would provide a more comprehensive picture. Third, although we focused on genes that exhibit significant changes in neonates compared with adults, genes with less pronounced differential expression in neonates may also be relevant. Finally, the underlying mechanisms of the identified hub genes remain uncertain, emphasizing the need for future studies.
Conclusions
We compared microglial transcriptomic alterations between neonatal and adult mice at 3 days post-SCI. Bioinformatics analysis identified 61 microglia-associated genes implicated in SCI. Furthermore, we investigated transcription factors, miRNAs, and potential drug targets associated with these genes. Subsequently, we selected 11 hub genes, including Chek1, RRM2, Lyve1, Mboat1, Clec4a3, Ccnd1, Cdk6, Zeb1, Igf1, Pparg and Cd163, from the 61 genes for further validation. These findings provide novel insights into neonatal microglial therapy for SCI and will contribute to the identification of novel therapeutic targets for this condition.
Data availability
The datasets analysed during the current study are available in the [GEO] repository, [https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE150871, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE113566]18,101. The rest of data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Bradbury, E. J. & Burnside, E. R. Moving beyond the glial Scar for spinal cord repair. Nat. Commun. 10 (1). https://doi.org/10.1038/s41467-019-11707-7 (2019).
Xiao, S. et al. The Syvn1 inhibits neuronal cell ferroptosis by activating Stat3/Gpx4 axis in rat with spinal cord injury. Cell Prolif. n/a (n/a), e13658. https://doi.org/10.1111/cpr.13658 (2024).
Wang, C. G. et al. A bioactive injectable self-healing anti-inflammatory hydrogel with ultralong extracellular vesicles release synergistically enhances motor functional recovery of spinal cord injury. Bioactive Mater. 6 (8), 2523–2534. https://doi.org/10.1016/j.bioactmat.2021.01.029 (2021).
Huang, H. Y. et al. Clinical neurorestorative therapeutic guidelines for spinal cord injury (IANR/CANR version 2019). J. Orthop. Transl. 14–24. https://doi.org/10.1016/j.jot.2019.10.006 (2020).
Badhiwala, J. H. et al. The influence of timing of surgical decompression for acute spinal cord injury: a pooled analysis of individual patient data. Lancet Neurol. 20 (2), 117–126. https://doi.org/10.1016/S1474-4422(20)30406-3 (2021).
Yin, W. et al. Identification of Anoikis-Related genes in spinal cord injury: bioinformatics and experimental validation. Mol. Neurobiol. https://doi.org/10.1007/s12035-024-04121-8 (2024).
Sofroniew, M. V. Dissecting spinal cord regeneration. Nature 557 (7705), 343–350. https://doi.org/10.1038/s41586-018-0068-4 (2018).
Fehlings, M. G. et al. Efficacy and safety of Methylprednisolone sodium succinate in acute spinal cord injury: A systematic review. Global Spine J. 7, 2192–5682. https://doi.org/10.1177/2192568217706366 (2017).
Ikeda-Yorifuji, I. et al. Single-nucleus RNA sequencing identified cells with ependymal cell-like features enriched in neonatal mice after spinal cord injury. Neurosci. Res. 181, 1872–8111. https://doi.org/10.1016/j.neures.2022.04.006 (2022).
Bertels, H. et al. Neurotransmitter phenotype switching by spinal excitatory interneurons regulates locomotor recovery after spinal cord injury. Nat. Neurosci. 25 (5), 617–629. https://doi.org/10.1038/s41593-022-01067-9 (2022).
Zhou, B. et al. Facilitation of axon regeneration by enhancing mitochondrial transport and rescuing energy deficits. J. Cell Biol. 214 (1), 103–119. https://doi.org/10.1083/jcb.201605101 (2016).
He, Z. & Jin, Y. Intrinsic control of axon regeneration. Neuron 90 (3), 437–451. https://doi.org/10.1016/j.neuron.2016.04.022 (2016).
Tsujioka, H. & Yamashita, T. Comparison of gene expression profile of the spinal cord of sprouting-capable neonatal and sprouting-incapable adult mice. BMC Genom. 20, 1471–2164. https://doi.org/10.1186/s12864-019-5974-9 (2019).
Hilton, B. J. & Bradke, F. Can injured adult CNS axons regenerate by recapitulating development? Development 144 (19), 3417–3429. https://doi.org/10.1242/dev.148312 (2017).
Brennan, F. H. et al. Microglia coordinate cellular interactions during spinal cord repair in mice. Nat. Commun. 13 (1). https://doi.org/10.1038/s41467-022-31797-0 (2022).
Bellver-Landete, V. et al. Microglia are an essential component of the neuroprotective Scar that forms after spinal cord injury. Nat. Commun. 10, 2041 – 1723 . https://doi.org/10.1038/s41467-019-08446-0 (2019).
Fu, H. T. et al. Depletion of microglia exacerbates injury and impairs function recovery after spinal cord injury in mice. Cell Death Dis. 11 (7). https://doi.org/10.1038/s41419-020-2733-4 (2020).
Li, Y. et al. Microglia-organized scar-free spinal cord repair in neonatal mice. Nature 587 (7835), 613–618. https://doi.org/10.1038/s41586-020-2795-6 (2020).
Sun, C. et al. The dual role of microglia in neuropathic pain after spinal cord injury: detrimental and protective effects. Exp. Neurol. 370, 114570. https://doi.org/10.1016/j.expneurol.2023.114570 (2023).
Barrett, T. et al. NCBI GEO: archive for functional genomics data sets—update. Nucleic Acids Res. 41 (D1), D991. https://doi.org/10.1093/nar/gks1193 (2013).
Butler, A. et al. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat. Biotechnol. 36 (5), 411–420. https://doi.org/10.1038/nbt.4096 (2018).
Xie, J. M. et al. Tree visualization by one table (tvBOT): a web application for visualizing, modifying and annotating phylogenetic trees. Nucleic Acids Res. 51 (W1), W587. https://doi.org/10.1093/nar/gkad359 (2023).
Gu, Z., Eils, R. & Schlesner, M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 32 (18), 2847–2849. https://doi.org/10.1093/bioinformatics/btw313 (2016).
Yu, W. W. et al. Kir2.1-mediated membrane potential promotes nutrient acquisition and inflammation through regulation of nutrient transporters. Nat. Commun. 13 (1). https://doi.org/10.1038/s41467-022-31149-y (2022).
Zhou, Y. Y. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 2041 – 1723 . https://doi.org/10.1038/s41467-019-09234-6 (2019).
Szklarczyk, D. et al. The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 45 (D1), D362. https://doi.org/10.1093/nar/gkw937 (2017).
Shannon, P. et al. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 13 (11), 2498–2504. https://doi.org/10.1101/gr.1239303 (2003).
Sticht, C. et al. MiRWalk: an online resource for prediction of MicroRNA binding sites. PLos One 13 (10). https://doi.org/10.1371/journal.pone.0206239 (2018).
Zhou, G. Y. et al. NetworkAnalyst 3.0: a visual analytics platform for comprehensive gene expression profiling and meta-analysis. Nucleic Acids Res. 47 (W1), W234. https://doi.org/10.1093/nar/gkz240 (2019).
Landt, S. G. et al. ChIP-seq guidelines and practices of the ENCODE and ModENCODE consortia. Genome Res. 22 (9), 1813–1831. https://doi.org/10.1101/gr.136184.111 (2012).
Dai, W. et al. LPIN1 is a regulatory factor associated with immune response and inflammation in Sepsis. Front. Immunol. 13, 1664–3224. https://doi.org/10.3389/fimmu.2022.820164 (2022). (Electronic)).
Yoo, M. et al. DSigDB: drug signatures database for gene set analysis. Bioinformatics 31 (18), 3069–3071. https://doi.org/10.1093/bioinformatics/btv313 (2015).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44 (W1), W90. https://doi.org/10.1093/nar/gkw377 (2016).
Liu, K. et al. PTEN deletion enhances the regenerative ability of adult corticospinal neurons. Nat. Neurosci. 13 (9), 1075–1081. https://doi.org/10.1038/nn.2603 (2010).
Fan, J. M. et al. Critical role of checkpoint kinase 1 in spinal cord injury-induced motor dysfunction in mice. Int. Immunopharmacol. 138, 1878 – 1705 . https://doi.org/10.1016/j.intimp.2024.112521 (2024).
Trost, A. et al. Lymphatic and vascular markers in an optic nerve crush model in rat. Exp. Eye Res. 159, 1096–0007. https://doi.org/10.1016/j.exer.2017.03.003 (2017).
Wang, Y. et al. Protective effect of epigenetic Silencing of CyclinD1 against spinal cord injury using bone marrow-derived mesenchymal stem cells in rats. J. Cell. Physiol. 233 (7), 5361–5369. https://doi.org/10.1002/jcp.26354 (2018).
Yin, W. et al. Identification of Anoikis-Related genes in spinal cord injury: bioinformatics and experimental validation. Mol. Neurobiol. 61 (11), 8531–8543. https://doi.org/10.1007/s12035-024-04121-8 (2024).
Gu, J. et al. Nanog interact with CDK6 to regulates astrocyte cells proliferation following spinal cord injury. Biochem. Biophys. Res. Commun. 469 (4), 1097–1103. https://doi.org/10.1016/j.bbrc.2015.12.053 (2016).
Yan, X. T. et al. XIST accelerates neuropathic pain progression through regulation of miR-150 and ZEB1 in CCI rat models. J. Cell. Physiol. 233 (8), 6098–6106. https://doi.org/10.1002/jcp.26453 (2018).
Peng, W. et al. Targeted delivery of CD163 + macrophage-derived small extracellular vesicles RGD peptides promote vascular regeneration and stabilization after spinal cord injury. J. Controlled Release. 361, 1873–4995. https://doi.org/10.1016/j.jconrel.2023.08.025 (2023).
Kaser-Eichberger, A. et al. Expression of lymphatic markers in the adult rat spinal cord. Front. Cell. Neurosci. 10, 1662–5102. https://doi.org/10.3389/fncel.2016.00023 (2016). (Print)).
Anjum, A. et al. Spinal cord injury: pathophysiology, multimolecular interactions, and underlying recovery mechanisms. Int. J. Mol. Sci. 21 (20). https://doi.org/10.3390/ijms21207533 (2020).
Li, C. et al. Temporal and Spatial cellular and molecular pathological alterations with single-cell resolution in the adult spinal cord after injury. Signal. Transduct. Target. Ther. 7 (1), 65. https://doi.org/10.1038/s41392-022-00885-4 (2022).
Hellenbrand, D. J. et al. Inflammation after spinal cord injury: a review of the critical timeline of signaling cues and cellular infiltration. J. Neuroinflamm. 18 (1). https://doi.org/10.1186/s12974-021-02337-2 (2021).
Hu, X. et al. Spinal cord injury: molecular mechanisms and therapeutic interventions. Signal. Transduct. Target. Therapy 8 (1). https://doi.org/10.1038/s41392-023-01477-6 (2023).
Hilton, B. J., Moulson, A. J. & Tetzlaff, W. Neuroprotection and secondary damage following spinal cord injury: concepts and methods. Neurosci. Lett. 652, 3–10. https://doi.org/10.1016/j.neulet.2016.12.004 (2017).
Wheaton, B. J. et al. Identification of regenerative processes in neonatal spinal cord injury in the opossum (Monodelphis domestica): A transcriptomic study. J. Comp. Neurol. 529 (5), 969–986. https://doi.org/10.1002/cne.24994 (2021).
Hernández Borrero, L. J. & El-Deiry, W. S. Tumor suppressor p53: Biology, signaling pathways, and therapeutic targeting. Biochim. Biophys. Acta (BBA) - Rev. Cancer 1876(1), 188556. https://doi.org/10.1016/j.bbcan.2021.188556 (2021).
Lei, Y. Myricitrin decreases traumatic injury of the spinal cord and exhibits antioxidant and anti-inflammatory activities in a rat model via Inhibition of COX-2, TGF-β1, p53 and elevation of Bcl-2/Bax signaling pathway. Mol. Med. Rep. 16 (5), 7699–7705. https://doi.org/10.3892/mmr.2017.7567 (2017).
Li, C. J. et al. Microglia-Derived Exosomal microRNA-151-3p Enhances Functional Healing After Spinal Cord Injury by Attenuating Neuronal Apoptosis Regulating the p53/p21/CDK1 Signaling Pathway. Front. Cell Dev. Biol. 9, 2296-634X. https://doi.org/10.3389/fcell.2021.783017 (2022).
Li, D. et al. Dihydroorotate dehydrogenase regulates ferroptosis in neurons after spinal cord injury via the P53-ALOX15 signaling pathway. CNS Neurosci. Ther. 29 (7), 1923–1939. https://doi.org/10.1111/cns.14150 (2023).
Jiang, X. J., Stockwell, B. R. & Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22 (4), 266–282. https://doi.org/10.1038/s41580-020-00324-8 (2021).
Hao, J. et al. Mechanisms underlying the promotion of functional recovery by deferoxamine after spinal cord injury in rats. Neural Regeneration Res. 12 (6), 959–968. https://doi.org/10.4103/1673-5374.208591 (2017).
Xu, S. et al. CD73 alleviates GSDMD-mediated microglia pyroptosis in spinal cord injury through PI3K/AKT/Foxo1 signaling. Clin. Translational Med. 11 (1). https://doi.org/10.1002/ctm2.269 (2021).
Fruman, D. A. et al. The PI3K pathway in human disease. Cell 170 (4), 605–635. https://doi.org/10.1016/j.cell.2017.07.029 (2017).
Hou, Y. et al. Resveratrol provides neuroprotection by regulating the JAK2/STAT3/PI3K/AKT/mTOR pathway after stroke in rats. Genes Dis. 5 (3), 245–255. https://doi.org/10.1016/j.gendis.2018.06.001 (2018).
Wang, S., Dougherty, E. J. & Danner, R. L. PPARγ signaling and emerging opportunities for improved therapeutics. Pharmacol. Res. 111, 1096–1186. https://doi.org/10.1016/j.phrs.2016.02.028 (2016).
Schupp, M. & Lazar, M. A. Endogenous ligands for nuclear receptors: digging deeper. J. Biol. Chem. 285 (52), 40409–40415. https://doi.org/10.1074/jbc.R110.182451 (2010).
Li, B. & Mei, X. F. Naringin May promote functional recovery following spinal cord injury by modulating microglial polarization through the PPAR-γ/NF-κB signaling pathway. Brain Res. 1821, 148563. https://doi.org/10.1016/j.brainres.2023.148563 (2023).
Park, S. W. et al. Thiazolidinedione class of peroxisome Proliferator-Activated receptor γ agonists prevents neuronal damage, motor dysfunction, Myelin loss, neuropathic pain, and inflammation after spinal cord injury in adult rats. J. Pharmacol. Exp. Ther. 320 (3), 1002. https://doi.org/10.1124/jpet.106.113472 (2007).
Zhao, X. et al. Neuronal PPARgamma deficiency increases susceptibility to brain damage after cerebral ischemia. J. Neurosci. 29 (19), 6186–6195. https://doi.org/10.1523/JNEUROSCI.5857-08.2009 (2009).
Ding, H. et al. Searching for differentially expressed proteins in spinal cord injury based on the proteomics analysis. Life Sci. 242, 117235. https://doi.org/10.1016/j.lfs.2019.117235 (2020).
Paterniti, I. et al. Molecular evidence for the involvement of PPAR-delta and PPAR-gamma in anti-inflammatory and neuroprotective activities of palmitoylethanolamide after spinal cord trauma. J. Neuroinflammation. 10, 1742–2094. https://doi.org/10.1186/1742-2094-10-20 (2013).
Hardie, D. G., Ross, F. A. & Hawley, S. A. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 13 (4), 251–262. https://doi.org/10.1038/nrm3311 (2012).
Chen, M. et al. The signaling pathways regulating NLRP3 inflammasome activation. Inflammation 44 (4), 1229–1245. https://doi.org/10.1007/s10753-021-01439-6 (2021).
Hu, H. S. et al. Zinc regulates glucose metabolism of the spinal cord and neurons and promotes functional recovery after spinal cord injury through the AMPK signaling pathway. Oxid. Med. Cell. Longev. 2021, 1942 – 0994 . https://doi.org/10.1155/2021/4331625 (2021).
Wu, J., Stoica, B. A. & Faden, A. I. Cell cycle activation and spinal cord injury. Neurotherapeutics 8 (2), 221–228. https://doi.org/10.1007/s13311-011-0028-2 (2011).
Wu, J. et al. Delayed cell cycle pathway modulation facilitates recovery after spinal cord injury. Cell. Cycle. 11 (9), 1782–1795. https://doi.org/10.4161/cc.20153 (2012).
Malumbres, M. & Barbacid, M. Cell cycle, CDKs and cancer: a changing paradigm. Nat. Rev. Cancer. 9 (3), 153–166. https://doi.org/10.1038/nrc2602 (2009).
Li, N. et al. Vitamin D promotes remyelination by suppressing c-Myc and inducing oligodendrocyte precursor cell differentiation after traumatic spinal cord injury. Int. J. Biol. Sci. 18 (14), 5391–5404. https://doi.org/10.7150/ijbs.73673 (2022).
Liu, J. et al. Exosomes derived from LncRNA TCTN2-Modified mesenchymal stem cells improve spinal cord injury by miR-329-3p/IGF1R Axis. J. Mol. Neurosci. 72 (3), 482–495. https://doi.org/10.1007/s12031-021-01914-7 (2022).
Zhang, Q. et al. miR–222-3p reduces neuronal cell apoptosis and alleviates spinal cord injury by inhibiting Bbc3 and Bim. Neurosci. Res. 188, 1872–8111. https://doi.org/10.1016/j.neures.2022.10.008 (2023).
Jiang, D. et al. Neuron-derived exosomes-transmitted miR-124-3p protect traumatically injured spinal cord by suppressing the activation of neurotoxic microglia and astrocytes. J. Nanobiotechnol. 18 (1), 105. https://doi.org/10.1186/s12951-020-00665-8 (2020).
Li, R. et al. Bone marrow mesenchymal stem cell-derived Exosomal microRNA-124-3p attenuates neurological damage in spinal cord ischemia-reperfusion injury by downregulating Ern1 and promoting M2 macrophage polarization. Arthritis Res. Ther. 22 (1), 75. https://doi.org/10.1186/s13075-020-2146-x (2020).
Jiang, M. et al. MicroRNA-124-3p attenuates the development of nerve injury-induced neuropathic pain by targeting early growth response 1 in the dorsal root ganglia and spinal dorsal Horn. J. Neurochem. 158 (4), 928–942. https://doi.org/10.1111/jnc.15433 (2021).
He, N. et al. Resveratrol suppresses microglial activation and promotes functional recovery of traumatic spinal cord via improving intestinal microbiota. Pharmacol. Res. 183, 106377. https://doi.org/10.1016/j.phrs.2022.106377 (2022).
Park, S. et al. Antibacterial nanocarriers of Resveratrol with gold and silver nanoparticles. Mater. Sci. Engineering: C. 58, 1160–1169. https://doi.org/10.1016/j.msec.2015.09.068 (2016).
Yang, C. M. et al. Resveratrol inhibits BK-induced COX-2 transcription by suppressing acetylation of AP-1 and NF-κB in human rheumatoid arthritis synovial fibroblasts. Biochem. Pharmacol. 132, 77–91. https://doi.org/10.1016/j.bcp.2017.03.003 (2017).
Zhao, H. et al. Resveratrol protects against spinal cord injury by activating autophagy and inhibiting apoptosis mediated by the SIRT1/AMPK signaling pathway. Neuroscience 348, 241–251. https://doi.org/10.1016/j.neuroscience.2017.02.027 (2017).
Lagouge, M. et al. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127 (6), 1109–1122. https://doi.org/10.1016/j.cell.2006.11.013 (2006).
Jiang, X. et al. Neuroprotection against spinal cord Ischemia–Reperfusion injury induced by different ischemic postconditioning methods: roles of phosphatidylinositol 3-Kinase–Akt and extracellular Signal–regulated kinase. Anesthesiology 111 (6), 1197–1205. https://doi.org/10.1097/ALN.0b013e3181bf1d93 (2009).
Bai, L. et al. Upregulation of chemokine CXCL12 in the dorsal root ganglia and spinal cord contributes to the development and maintenance of neuropathic pain following spared nerve injury in rats. Neurosci. Bull. 32 (1), 27–40. https://doi.org/10.1007/s12264-015-0007-4 (2016).
Shvetcov, A. et al. The neuroprotective effects of Estrogen and Estrogenic compounds in spinal cord injury. Neurosci. Biobehav Rev. 146, 1873–7528. https://doi.org/10.1016/j.neubiorev.2023.105074 (2023).
Bagheri, M. et al. Amyloid beta(1–40)-induced astrogliosis and the effect of genistein treatment in rat: a three-dimensional confocal morphometric and proteomic study. PLoS One 8 (10), e. https://doi.org/10.1371/journal.pone.0076526 (2013).
Ji, G. et al. Genistein suppresses LPS-induced inflammatory response through inhibiting NF-kappaB following AMP kinase activation in RAW 264.7 macrophages. PLoS One 7 (12), e53101. https://doi.org/10.1371/journal.pone.0053101 (2012).
Ismail, M. et al. Genistein loaded nanofibers protect spinal cord tissue following experimental injury in rats. Biomedicines 6 (4). https://doi.org/10.3390/biomedicines6040096 (2018).
Gu, C. et al. CHEK1 and circCHEK1_246aa evoke chromosomal instability and induce bone lesion formation in multiple myeloma. Mol. Cancer 20 (1), 84. https://doi.org/10.1186/s12943-021-01380-0 (2021).
Yamashita, R. et al. Induction of cellular senescence as a late effect and BDNF-TrkB signaling-mediated ameliorating effect on disruption of hippocampal neurogenesis after developmental exposure to lead acetate in rats. Toxicology 456, 152782. https://doi.org/10.1016/j.tox.2021.152782 (2021).
Khan, P. et al. MicroRNA-1 attenuates the growth and metastasis of small cell lung cancer through CXCR4/FOXM1/RRM2 axis. Mol. Cancer. 22 (1), 1. https://doi.org/10.1186/s12943-022-01695-6 (2023).
Yang, Y. et al. RRM2 protects against ferroptosis and is a tumor biomarker for liver cancer. Cancer Cell. Int. 20 (1), 587. https://doi.org/10.1186/s12935-020-01689-8 (2020).
Lindblom, R. P. F. et al. Genetic variability in the rat aplec C-type lectin gene cluster regulates lymphocyte trafficking and motor neuron survival after traumatic nerve root injury. J. Neuroinflamm. 10 (1), 831. https://doi.org/10.1186/1742-2094-10-60 (2013).
Kabadi, S. V. et al. Cyclin D1 gene ablation confers neuroprotection in traumatic brain injury. J. Neurotrauma. 29 (5), 813–827. https://doi.org/10.1089/neu.2011.1980 (2012).
Liang, D. et al. Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones. Cell 186 (13), 2748–2764. https://doi.org/10.1016/j.cell.2023.05.003 (2023).
Sex hormone signaling suppresses ferroptosis via phospholipid remodeling. Cancer Discov. 13(8), 1759. https://doi.org/10.1158/2159-8290.CD-RW2023-091 (2023).
Allahdadi, K. J. et al. IGF-1 overexpression improves mesenchymal stem cell survival and promotes neurological recovery after spinal cord injury. Stem Cell Res. Ther. 10 (1), 146. https://doi.org/10.1186/s13287-019-1223-z (2019).
Craig, A. et al. Prospective study of the occurrence of psychological disorders and comorbidities after spinal cord injury. Arch. Phys. Med. Rehabil. 96 (8), 1426–1434. https://doi.org/10.1016/j.apmr.2015.02.027 (2015).
Wu, J. et al. Isolated spinal cord contusion in rats induces chronic brain neuroinflammation, neurodegeneration, and cognitive impairment. Involvement of cell cycle activation. Cell. Cycle. 13 (15), 2446–2458. https://doi.org/10.4161/cc.29420 (2014).
Jure, I. et al. IGF1 gene therapy reversed cognitive deficits and restored hippocampal alterations after chronic spinal cord injury. Mol. Neurobiol. 58 (12), 6186–6202. https://doi.org/10.1007/s12035-021-02545-0 (2021).
Yang, S. et al. TREM2-IGF1 mediated glucometabolic enhancement underlies microglial neuroprotective properties during ischemic stroke. Adv. Sci. 11 (10). https://doi.org/10.1002/advs.202305614 (2024).
Zhou, X. et al. Microglia and macrophages promote corralling, wound compaction and recovery after spinal cord injury via Plexin-B2. Nat. Neurosci. 23 (3), 337–350. https://doi.org/10.1038/s41593-020-0597-7 (2020).
Kanehisa, M. Toward Understanding the origin and evolution of cellular organisms. Protein Sci. 28 (11), 1947–1951. https://doi.org/10.1002/pro.3715 (2019).
Kanehisa, M. et al. KEGG: biological systems database as a model of the real world. Nucleic Acids Res. 1362–4962. https://doi.org/10.1093/nar/gkae909 (2024). (Electronic)).
Kanehisa, M. et al. KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res. 51 (D1), D587. https://doi.org/10.1093/nar/gkac963 (2023).
Kanehisa, M. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28 (1), 27–30. https://doi.org/10.1093/nar/28.1.27 (2000).
Acknowledgements
Thanks to the public databases for providing us with data, and thanks to the developers of R software and R packages for their contributions and convenience.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82102558), the Shandong Province Natural Science Foundation (ZR2020QH117), the Shandong Provincial Medical and Health Science and Technology Project(202407020962)and the China Postdoctoral Science Foundation (2021M691690).
Author information
Authors and Affiliations
Contributions
Q. J and S.X performed the experiments and drafted the manuscript. H.F and D.H conceived the project and designed the experiments. Z.R and L.L contributed to the performed the data analysis. X.W and X.P contributed to the mRNA analysis. W.S contributed to the establishment and intervention of animal models. T.Y and C.Q helped to interpret the data. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All procedures performed in studies involving animals were approved by the Laboratory Animal Welfare Ethics Committee of Qingdao University.
Consent for publication
All authors approved the manuscript and gave their consent for submission and publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Jiang, Q., Xue, S., Pan, X. et al. Differential changes in the microglial transcriptome between neonatal and adult mice after spinal cord injury. Sci Rep 15, 13708 (2025). https://doi.org/10.1038/s41598-025-98429-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-025-98429-7