Abstract
Metastasis to the cerebrospinal-fluid-filled leptomeninges, or leptomeningeal metastasis, represents a fatal complication of solid tumours1. Multimodal analyses of clinical specimens reveal substantial inflammatory infiltrate in leptomeningeal metastases with enrichment of IFNγ and resulting downstream signalling. Here, to investigate and overcome this futile anti-tumour response within the leptomeninges, we developed syngeneic lung cancer, breast cancer and melanoma leptomeningeal-metastasis mouse models. We show that transgenic host mice lacking IFNγ or its receptor fail to control the growth of leptomeningeal metastases growth. Leptomeningeal overexpression of Ifng through a targeted adeno-associated-virus-based system controls cancer cell growth independent of adaptive immunity. Using a suite of transgenic hosts, we demonstrate that leptomeningeal T cells generate IFNγ to actively recruit and activate peripheral myeloid cells, generating a diverse spectrum of dendritic cell subsets. Independent of antigen presentation, migratory CCR7+ dendritic cells orchestrate the influx, proliferation and cytotoxic action of natural killer cells to control cancer cell growth in the leptomeninges. This study identifies unique, leptomeninges-specific IFNγ signalling and suggests an immune-therapeutic approach against tumours within this space.
Similar content being viewed by others
Main
Metastasis, or the spread of cancer to distant anatomical sites, requires cancer cells to enter and thrive within microenvironments unlike those of the primary tumour. In parallel, immune cells migrate throughout the organism and enter the same microenvironments as a counteroffensive, controlling disseminated malignant cell growth. This balance may tip in favour of cancer cell growth when anti-tumour immune cell populations are excluded, as is the case for the majority of metastases to the central nervous system (CNS)2. An important exception is that of leptomeningeal metastasis (LM). The leptomeninges—cerebrospinal fluid (CSF)-filled membranes—encase the CNS. Cancer cell entry into the leptomeningeal space, or LM, provokes a robust inflammatory response3,4,5 clinically reminiscent of infectious meningitis6,7,8. Despite the presence of abundant immune cells and intense inflammation, leptomeningeal cancer cells persist and even thrive, a consequence of inflammation-mediated transcriptional changes within these cancer cells3,6. How and why this immune infiltrate fails to control cancer cell growth remains unclear. Previous research revealed non-canonical transcriptional and functional changes in macrophages in LM, suggesting that other immune cells within this anatomical compartment may also behave atypically6. Moreover, the levels of inflammatory cytokines in the leptomeningeal space do not reflect the plasma levels, consistent with both intrathecal cytokine generation and alternative regulatory system(s) within this space9. Formal investigations of this complex, anatomically site-specific call and response between immune cells and cancer cells have remained incomplete owing to lack of immunocompetent mouse models3,10 and piecemeal computational approaches that do not encompass the entirety of cellular and humoral signalling within this anatomic compartment. To examine the leptomeningeal anti-tumour response in an unbiased manner, we captured and comprehensively molecularly dissected the immune response to LM in both human disease and immunocompetent mouse models at the cellular and humoral levels.
LM generates inflammation
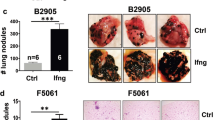
LM is a uniformly fatal neurological consequence of cancer1. Although any malignancy can lead to LM, it most commonly results from breast, lung and melanoma primaries11. Cancer cells enter into the leptomeningeal space accompanied by a host of leukocytes, as can be appreciated by classical techniques including CSF cytospin (Fig. 1a). To understand this leukocytic pleocytosis at a molecular level, we profiled CSF collected from patients with breast and lung cancer with (n = 5) and without (n = 3) LM using the 10x platform, collecting single-cell transcriptomes and assigning cell identity by clustering and marker gene expression (Fig. 1b and Supplementary Fig. 1). In the absence of LM, the CSF is nearly acellular and contains predominantly CD4+ T cells (Fig. 1a–c). CSF from patients with LM was pleocytic and contained cells from a spectrum of lymphoid and myeloid lineages (Supplementary Fig. 1). To assess the molecular hallmarks of CSF pleocytosis and capture cell-to-cell communication, we subjected CSF from patients with breast cancer, lung cancer and melanoma primaries, with and without LM, to targeted proteomic analysis using a proximity extension assay11 (Extended Data Fig. 1a–c and Supplementary Tables 1–3). In the presence of LM, CSF demonstrated a robust influx of soluble inflammatory ligands; 15 of these molecules, notably IFNγ, were shared across the 3 tumour types (Extended Data Fig. 1d). We extended this cohort to include patients with a wide variety of solid tumour primaries and confirmed consistent elevation of CSF IFNγ in the presence of LM (Fig. 1d). Examining clinical outcomes, we find that elevated CSF IFNγ levels at diagnosis were associated with improved overall survival, as well as increased abundance of leptomeningeal lymphocytes and myeloid cells (Fig. 1e and Extended Data Fig. 1e). We confirmed these observations in an independent cohort of patients using a high-sensitivity enzyme-linked immunosorbent assay (ELISA) for IFNγ (Extended Data Fig. 1f–h). IFNγ is well known to provoke both pro-tumorigenic and tumour-suppressive actions in a context-dependent manner; the presence of inflammatory and anti-inflammatory signals in cancer-infiltrated leptomeninges suggested a dense signalling network that is not clearly consistent with canonical pathways observed outside the CNS. We therefore pursued formal identification of downstream leptomeningeal effectors of IFNγ and the functional consequences of their activation.
a, Representative images of Giemsa-stained cytospins from patients with cancer without (top) and with (bottom) LM; major cell populations are indicated. Ly, lymphocytes; MM, monocytes and macrophages; cc, cancer cells. n = 5 per group. Scale bars, 20 μm. b, Uniform manifold approximation and projection (UMAP) projection of human CSF immune cell types and cancer cells, isolated from patients with cancer without (n = 3 patients and n = 1,196 cells) and with (n = 5 patients and n = 16,022 cells) LM. LM+ samples were retrieved from Gene Expression Omnibus (GEO) GSE150660 and coloured by cell type6. An experimental overview, cell type annotations and quality-control plots are provided in Supplementary Fig. 1 and the Methods. c, Embedding density plots from patients without or with LM, projecting the relative cell type abundance per condition onto the UMAP. d, Relative CSF IFNγ levels in patients with cancer with or without LM from a wide array of solid tumours, as determined by proximity extension assay. NPX, normalized protein expression. See also Extended Data Fig. 1. e, Post-LM diagnosis survival in relation to CSF IFNγ levels at diagnosis. Statistical analysis was performed using the log-rank test.
IFNγ regulates LM growth
To enable these mechanistic studies, we used iterative in vivo selection to generate six immunocompetent mouse leptomeningeal metastatic lines (LeptoM) on two genetic backgrounds3 (Extended Data Fig. 2a). These cell lines, subpopulations of the founding parental line, are phenotypically and transcriptomically distinct from their parental or brain parenchymal-tropic counterparts (Extended Data Fig. 2b–m). These LeptoM models faithfully recapitulate key histological and oncological features of human LM, including CSF pleocytosis (Fig. 2a), brisk pace of illness (Extended Data Fig. 2b–m) and induction of a systemic immune response that fails to control leptomeningeal tumours (Extended Data Fig. 3). To capture the leptomeningeal immune infiltrate at the systems level, we performed proteogenomic analysis of mouse leptomeningeal immune infiltrate with 198 barcoded antibodies targeting cell-surface epitopes and non-targeting isotype controls coupled with single-cell RNA-sequencing (scRNA-seq) on the 10x platform (scCITE-seq)12. This approach enables granular identification of immune cell subtypes and their origins with commonly used cytometric markers, guiding the identification of populations of interest on the basis of protein and mRNA expression. These mouse models mimic human CSF cellular composition in the setting of LM—a substantial influx of leukocytes is observed, evenly split between myeloid and lymphoid populations (Fig. 2b,c and Supplementary Fig. 2; compare Fig. 1b,c and Supplementary Fig. 1). In the absence of LM, human CSF is T cell predominant; mouse leptomeninges are populated by B cells and neutrophils. Despite species-specific differences in the absence of malignancy, the presence of LM drives the CSF cellular composition to a common, lymphocyte-dominant and to some extent also myeloid-rich pleocytosis, independent of vertebrate host.
a, Representative images of haematoxylin-stained cytospins from vehicle-injected (top) and E0771 LeptoM-injected (bottom) mice. n = 3 per group. Scale bars, 50 μm. See also Extended Data Fig. 2. b, UMAP analysis of leptomeningeal cellular material from vehicle- and LLC LeptoM-injected mice 2 weeks after inoculation, after single-cell proteogenomic profiling using 10x CITE-seq. n = 7,528 (vehicle injected) and n = 19,534 (LLC LeptoM-injected) cells, n = 6 mice per group. See also Supplementary Figs. 2 and 3. c, Embedding density plots from LM− and LM+ mice, projecting the relative cell type abundance per condition onto the UMAP. d, Cytometry bead array quantification of CSF IFNγ from naive or LeptoM-bearing mice. e, Representative leptomeningeal tissue sections were stained with haematoxylin and eosin (H&E). Scale bars, 100 μm. The box plot shows the brain surface area covered with pigmented B16 LeptoM cells delivered intracisternally into C57BL/6 Ifng-proficient and Ifng-deficient mice, 2 weeks after injection. f, In vivo radiance of LLC LeptoM cells delivered intracisternally into mice with WT T cells and mice with ΔIfng T cells, quantified 2 weeks after injection. g, In vivo radiance of LLC LeptoM cells delivered intracisternally into mice with WT NK cells and mice with ΔIfng NK cells, quantified 2 weeks after injection. h, Representative leptomeningeal tissues were stained with H&E. The box plot shows the brain surface area covered with pigmented B16 LeptoM cells delivered intracisternally into C57BL/6 Ifngr1−/− and Ifngr1+/+ mice, 2 weeks after injection. The basilar meninges of these animals is shown. Scale bars, 100 μm (left) and 5 mm (right). i, Survival of mice after control LeptoM cells (sglacz, n = 10) and two Ifngr2-deficient B16 LeptoM clones (sg1, n = 10 and sg2, n = 9) were delivered intracisternally into C57BL/6 mice. Median overall survival: mOS sglacz (18 days), sg1 (15 days), sg2 (18 days). j, Schematic showing that the leptomeningeal T cells are the dominant generator of IFNγ-mediated cancer cell elimination.
We investigated cellular responses to leptomeningeal IFNγ signalling13. Mirroring human disease (Fig. 1d), we detected elevated CSF levels of IFNγ in mice with LM, compared with vehicle-injected animals (Fig. 2d). To identify the source of IFNγ, we subjected CSF collected from mice with and without LM to flow cytometry assessment of IFNγ production (Extended Data Fig. 4a). We found that leptomeningeal T and natural killer (NK) cells produce IFNγ. In parallel, we queried our human and mouse single-cell atlases for IFNG transcripts. In both species, T cells and NK cells produce IFNγ (Extended Data Fig. 4b). As IFNγ binding to its cognate receptors triggers a signalling cascade that results in phosphorylation of STAT1 (pSTAT1)14, we assessed the levels of pSTAT1 in leptomeningeal immune infiltrates using flow cytometry. We detected increased levels of pSTAT1 in mouse leptomeningeal dendritic cells (DCs), monocytes and macrophages and T cells, but not NK cells (Extended Data Fig. 4c). Validating these findings in human disease, we found enrichment of the IFNγ gene signature in human DCs and monocytes and macrophages and, to a lesser extent, T cells (Extended Data Fig. 4d,e). While depletion of IFNγ and IFNγ-producing cells did not alter leptomeningeal pSTAT1 levels, it substantially decreased the abundance of leptomeningeal DCs and NK cells (Extended Data Fig. 2f–k). These results support a model whereby leptomeningeal monocytes and macrophages, DCs and T cells respond to IFNγ generated by leptomeningeal T and NK cells.
To molecularly dissect this response, we used transgenic host mice lacking either Ifng or its receptor Ifngr1, resulting in whole-body impairment of IFNγ signalling15,16. In the absence of IFNγ, the host leptomeningeal anti-tumour response was impaired, and leptomeningeal tumours grew unchecked (Fig. 2e and Extended Data Fig. 5a,b). To determine the source of IFNγ, we used transgenic hosts. Selective ablation of IFNγ generation by either T or NK cells demonstrated that T cells generate the majority of the IFNγ responsible for the anti-tumour response (Fig. 2f,g). Interruption of IFNγ signalling in whole-body Ifngr1-knockout mice led to uncontrolled cancer cell growth in the leptomeninges (Fig. 2h and Extended Data Fig. 5c–e). This effect was not observed when LeptoM cells were orthotopically implanted into primary tumour sites or subcutaneously (Extended Data Fig. 6a–f), consistent with a leptomeninges-specific role for IFNγ mediated anti-tumour response. After haematogenous dissemination, Ifngr1-deficient hosts had a higher incidence of metastases only in the thorax (Extended Data Fig. 6g–j).
In a context-dependent manner, IFNγ may either promote or inhibit cancer growth17. This can be the result of a direct IFNγ signal to the cancer cell or indirect signalling to the tumour microenvironment. To investigate direct IFNγ action on cancer cells, we knocked out the Ifngr2 subunit of IFNγ receptor using CRISPR–Cas9. In contrast to the control clones, these knockout lines were unable to propagate canonical IFNγ upregulation of MHC class I on the cell surface (Extended Data Fig. 7a–c). The lack of Ifngr2 in these cells did not alter their growth in vitro (Extended Data Fig. 7d–f) or in vivo, and the outcomes were cell clone specific (Extended Data Fig. 7g,h). We concluded that cancer-intrinsic IFNγ signalling is not required for cancer cell survival in the leptomeninges. IFNγ therefore mediates leptomeningeal cancer cell growth through effects on the microenvironment (Fig. 2j). As knockout of host IFNγ promoted cancer cell growth, we pursued a complementary add-back strategy with intracisternal introduction of recombinant mouse IFNγ. While LeptoM cancer cells demonstrated the ability to receive IFNγ signals (Extended Data Fig. 8a–c), this did not affect in vitro proliferation (Extended Data Fig. 7d–f). However, in vivo, addition of IFNγ suppressed cancer cell growth within the leptomeninges (Extended Data Fig. 8g–j), suggesting an interplay between IFNγ and other cells in this inflammatory microenvironment.
Innate immunity enacts anti-LM response
To identify downstream IFNγ effectors in the context of LM, we engineered an experimental system that enables manipulation of CSF composition without frequent anaesthesia or injection of foreign agents into the leptomeninges. We constructed an adeno-associated virus (AAV)-based system to induce the expression of exogenous Ifng or a control gene, eGFP, in the mouse leptomeninges18,19 (Fig. 3a and Extended Data Fig. 9a,b). Using this technique, overexpressed leptomeningeal IFNγ resulted in substantial control of leptomeningeal metastatic cancer cell growth in all six syngeneic LeptoM models; overexpressed eGFP did not (Fig 3b,c and Extended Data Fig. 9c–l). This overexpression system did not result in neurodegeneration or neuroinflammation, as has been observed with type I interferons20. We observed a normal profile of astrocytes lining the ventricular space, without apparent activation of parenchymal microglia, depletion of neural progenitors, change in neuronal tract distribution or change in mature cortical neuron numbers (Extended Data Fig. 10a–f). Although the overall number of leptomeningeal leukocytes did not change, we noticed an increased proportion of leptomeningeal T cells, NK cells and DCs after Ifng overexpression (Extended Data Fig. 10g,h). Similar to earlier reports, we detected a decrease in the immature oligodendrocyte population in the corpus callosum21,22. This was not reflected in the cortical and subcortical layers, in which we detected a minor decrease in differentiated, CNPase-positive cortical and subcortical oligodendrocytes (Extended Data Fig. 10i,j). Leptomeninges-specific overexpression of IFNγ therefore does not substantially impair brain physiology during our experimental timeframe.
a, Schematics of the simplified experimental strategy to generate leptomeningeal eGFP or Ifng overexpression (Methods and Extended Data Fig. 9). b, Representative leptomeningeal tissue sections were stained with H&E. Scale bars, 100 μm. The box plot illustrates the in vivo radiance of lung-cancer-derived LLC LeptoM cells delivered intracisternally into C57BL/6-Tyrc-2 mice overexpressing eGFP or Ifng in the leptomeninges, quantified 2 weeks after injection. c, Survival of LLC LeptoM-bearing C57BL/6-Tyrc-2 mice overexpressing eGFP or Ifng in the leptomeninges. Statistical analysis was performed using the log-rank test. d, In vivo radiance of LLC LeptoM cells delivered intracisternally into NSG mice overexpressing eGFP or Ifng in the leptomeninges, quantified 2 weeks after injection. NSG, non-obese, diabetic, severe combined immunodeficient, Il2rgnull mice. e, In vivo radiance of LLC LeptoM cells delivered intracisternally into RAG1-deficient animals overexpressing eGFP or Ifng in the leptomeninges, quantified 2 weeks after injection. f, Representative immunofluorescence image of brain tissue from cancer-naive mice overexpressing eGFP or Ifng in the leptomeninges, stained for IBA1+ myeloid cells. Scale bars, 50 μm. Quantification of IBA1+ cells in the ventricular choroid plexuses is shown. g, In vivo radiance of LLC LeptoM cells delivered intracisternally into mice with WT myeloid cells and mice with ΔIfngr1 myeloid cells and quantified 2 weeks after injection. Myeloid LysM-driven cre is expressed predominantly in monocytes, macrophages and neutrophils.
With this tool in hand, we set out to identify the microenvironmental cell population(s) that are responsible for IFNγ-dependent cancer control in the leptomeninges. We hypothesized that the anti-cancer effect of IFNγ was secondary to the immune system. We therefore established our leptomeningeal IFNγ overexpression system in fully immunodeficient NSG mice, and completely eliminated IFNγ’s anti-cancer activity (Fig. 3d and Extended Data Fig. 11a–c). IFNγ is well-known to positively regulate antigen presentation14. We next investigated the role that antigen presentation might have in IFNγ’s anti-tumour effect in the leptomeninges. We used Rag1-deficient animals (which are incapable of antigen presentation) as hosts, and observed that IFNγ’s anti-tumour effect was preserved, indicating that IFNγ’s anti-tumour function is independent of antigen presentation in the leptomeninges (Fig. 3e and Extended Data Fig. 11d–f). We therefore turned our attention to the innate branch of the immune system, focusing on IBA1+ monocytes and macrophages—well-known IFNγ effectors14. Overexpression of Ifng resulted in the accumulation of IBA1+ myeloid cells in the choroid plexus (Fig. 3f), an interface between the periphery and the leptomeninges that functions as a gateway for immune cell entry23,24. To specifically manipulate monocytes, macrophages and neutrophils in the host mice, we used the LysM-driven, myeloid-cell-specific cre driver. Conditional Ifngr1 deletion in myeloid cells did not result in impaired tumour growth, suggesting that these cell types are dispensable for IFNγ’s anti-cancer activity in LM (Fig. 3g). IFNγ-mediated leptomeningeal tumour control is therefore dependent on the immune system, but independent of antigen presentation, adaptive immunity, monocytes and macrophages, and neutrophils. We next addressed DCs, an IFNγ effector cell type spared from LysM-cre-mediated Ifngr1 deletion25.
DCs orchestrate LM response
Conventional DCs (cDCs) are a phagocytic myeloid immune cell lineage that can propagate IFNγ response, typically through antigen presentation26. However, their function in an antigen-independent setting is less explored. To specifically deplete the cDC lineage in the mice and clarify the functional role of these cells in LM, we took advantage of two transgenic systems expressing human diphtheria toxin receptor (DTR). In the first strain, DTR is expressed under the control of endogenous mouse Zbtb4627. Within the haematopoietic compartment, Zbtb46 expression is restricted to cDCs, type 3 innate lymphoid cells and Thetis cells (the latter two are not detected in our system)28,29. However, Zbtb46 is also expressed by other non-immune cell types, including endothelial cells. To avoid consequences related to systemic depletion of Zbtb46-expressing cells, we generated bone marrow chimeras. We infused lethally irradiated wild-type (WT) recipient mice with bone marrow from WT or Zbtb46-DTR animals. In these mice, diphtheria toxin (DTx) eliminates Zbtb46-expressing mature and progenitor cDCs (Extended Data Fig. 11g–i) while retaining the normal function of other, non-haematopoietic cell types. Depletion of cDCs in the Zbtb46-DTR system increased leptomeningeal, but not extracranial, tumour growth (Fig. 4a and Extended Data Fig. 11j). In a second, complementary approach, we depleted DCs by targeting Clec9a-expressing cells. This approach ablates both cDC1 and cDC2 populations, as Clec9a is expressed by the common DC progenitor30. As in the Zbtb46-targeted system, loss of the Clec9a-expressing DC populations increased leptomeningeal metastatic cancer cell growth (Fig. 4b and Extended Data Fig. 11k–m). To confirm that the cDC population is essential for IFNγ’s anti-tumour function, we overexpressed leptomeningeal Ifng or eGFP in Zbtb46-DTR bone marrow chimeras after bone marrow reconstitution and introduced cancer cells. Introduction of DTx into WT chimera hosts did not alter the activity of IFNγ; mice with ablated cDC populations demonstrated reduced IFNγ-dependent tumour control (Fig. 4c). Together, these experiments support a model whereby leptomeningeal DCs respond to IFNγ (Extended Data Fig. 4c–e) to mediate an anti-tumour action.
a, In vivo radiance of LLC LeptoM cells delivered intracisternally into Zbtb46-DTR (recipient C57BL/6-Tyrc-2) bone marrow chimeras, treated with DTx and measured 2 weeks after cancer cell injection (Methods and Extended Data Fig. 11). b, In vivo radiance of LLC LeptoM cells delivered intracisternally into Clec9acre and Clec9acreRosa26lsl-DTR mice treated with DTx, 2 weeks after injection. c, In vivo radiance of LLC LeptoM cells delivered intracisternally into Zbtb46-DTR (recipient C57BL/6-Tyrc-2) bone marrow chimeras overexpressing eGFP or Ifng into the leptomeninges, treated with DTx, 2 weeks after injection. d, t-Distributed stochastic neighbour embedding (t-SNE) of mouse leptomeningeal DC types from vehicle- (veh.) and LLC LeptoM-injected mice 2 weeks after inoculation, analysed using 10x CITE-seq (Methods and Extended Data Fig. 12). n = 7,566 cells pooled from 4 conditions, n = 6 animals per group. e, Surface protein and Ccr7 gene expression in DCs from the experiment in d. f, Gene expression trends associated with cDC2, CCR7+ identity and IFNγ signalling along the diffusion pseudotime axis, as determined using Palantir35. g, In vivo radiance of LLC LeptoM cells delivered intracisternally into mice with WT cDCs and mice with ΔIfngr1 cDCs, 2 weeks after injection. h, The proportion of mature leptomeningeal CCR7+ (CD11c+MHC-II+) DCs in LLC LeptoM-bearing Ifngr1-proficient and Ifngr1-deficient mice. i, T cell-derived IFNγ governs maturation of leptomeningeal cDC2 subset into mature, migratory CCR7+ DCs. j, UMAP of identity (left) and cell cycle phase prediction (right) in LLC LeptoM cancer cells expressing cytokeratin and CD63. n = 3,161 and n = 557 cells from mice overexpressing eGFP and Ifng in the leptomeninges, respectively; n = 6 mice per group. k, Quantification of the experiment in e (Methods and Extended Data Fig. 13i–k). l, Representative image of cleaved-caspase-3-stained (Cl. CASP3) E0771 LeptoM in mice overexpressing eGFP or Ifng in the leptomeninges. Scale bars, 50 μm; br., brain parenchyma. See also Extended Data Fig. 13l–n.
To capture the IFNγ response in the leptomeningeal space at the systems level, we isolated leptomeningeal cells from eGFP- and Ifng-overexpressing mice in the presence and absence of LM and profiled these cells using CITE-seq (Supplementary Fig. 3; total n = 24 mice from 4 conditions). We confirmed the presence of all classical DC populations: cDC1 and cDC2 cells, mature migratory CCR7+ DCs and plasmacytoid DCs within the leptomeninges (pDCs; Fig. 4d,e). Molecular profiling of DCs isolated from eGFP- and Ifng-overexpressing mice revealed marked similarities between mouse and human leptomeningeal DCs (Supplementary Fig. 4), as well as site-specific (leptomeningeal) imprinted expression patterns different from those observed within extracranial sites31,32. In the presence of cancer, or in the presence of IFNγ, cDC populations accumulate within the leptomeninges (Extended Data Fig. 12a–c). To address IFNγ-dependent relationships between these cDC populations, we queried our proteogenomic atlas. Outside the CNS, CCR7+ DCs can arise from both cDC1 and cDC2 populations33. However, the majority of leptomeningeal CCR7+ DCs retained the cDC2 surface expression profile, as detected by CITE-seq (Extended Data Fig. 11j). Given the leptomeninges-specific expression pattern, we approached this computationally. We first used CellRank to predict terminal cell states, without the need to indicate the initial cell34 (Extended Data Fig. 12f). This analysis identified the cDC2 cell population as the major contributors to the leptomeningeal CCR7+ DC pool; it also identified CCR7+ DCs as a product of cDC2 maturation (Extended Data Fig. 12g–h). We next reproduced the trajectory analyses using Palantir, modelling the cDC2–CCR7+ DC maturation axis35 (Extended Data Fig. 12i,j). We detected enrichment of IFNγ-associated genes as cells transition to CCR7+ DCs, consistent with IFNγ contribution to CCR7+ DC maturation from cDC2 cells26 (Fig. 4f). When we experimentally deleted Infgr1 in cDCs, we observed increased leptomeningeal tumour burden (Fig. 4g). Although whole-body Ifngr1-deficient mice had an abundance of cDCs comparable to Ifngr1-proficient counterparts, their maturation and cytokine production were impeded (Fig. 4h and Extended Data Fig. 13a,b). Both genetic and surface-protein-based lineage tracing confirmed that a minority of these CCR7+ DCs arose from the cDC1 subset (Extended Data Fig. 13c–e), suggesting that most of this mature population is cDC2 derived, as predicted computationally. Notably, in contrast to other meningeal immune cells of myeloid origin, these leptomeningeal cDCs originated predominantly from extracranial sources and not the proximal bone marrow36 (Extended Data Fig. 13f–h).
As the anti-tumour effect of leptomeningeal IFNγ is not mediated directly (Fig. 2i) and does not rely on antigen presentation (Fig. 4i), we next examined anti-tumour pathways that cDCs might affect, including cancer cell proliferation and death. Prediction of cancer cell cycling in our transcriptomic data revealed that cancer cells isolated from Ifng-overexpressing mice did not demonstrate defective proliferation (Fig. 4j,k and Extended Data Fig. 13i–k). However, immunofluorescence analysis of cancer cells in the leptomeninges identified elevated cleaved caspase activity in the Ifng-overexpressing mice, consistent with apoptotic cell death (Fig. 4l and Extended Data Fig. 13l–n). These results suggested that a cytotoxic immune population, supported by cDCs, restricts cancer cell expansion in the leptomeninges.
NK cells follow the DC lead
We next focused on NK cells, cytotoxic effectors adept at tumour cell killing37. Mouse leptomeninges contained naive, activated and proliferating NK cells. In the presence of cancer, a minor population of senescent NK cells was also present (Fig. 5a,b and Supplementary Fig. 5a–c). Human CSF demonstrated analogous populations of naive-like and activated-like NK cells (Supplementary Fig. 5d–g). Independent of cancer, leptomeningeal Ifng overexpression induced increased NK cell proliferation; this effect was retained in NK cells isolated from Ifng-overexpressing cancer-bearing animals (Fig. 5c).
a, t-SNE projection of leptomeningeal NK cells from vehicle- and LLC LeptoM-injected mice 2 weeks after inoculation, analysed using 10x CITE-seq. Total of n = 2,247 cells from 4 conditions, n = 6 mice per group (Methods). b, Cell-state-enriched NK surface protein expression from the experiment in a. N, naive; A, activated; P, proliferative; S, senescent-like. c, NK cell cycle prediction from the experiment in a (Methods). d, Selected CCR7+ DC ligand and NK cell receptor expression from the experiment in a, projected onto t-SNE plots (Methods). e, The relative abundance of CSF IL-12, IL-15 and IL-18 from patients without and with LM, as determined using targeted proteomics. Statistical analysis was performed using multiple t-tests. f, Human NK surface protein expression, as determined using single-cell transcriptomics. g, NK cell survival in the CSF of patients without and with LM. Two independent experiments (Methods). h, NK cell survival in human LM− CSF with or without recombinant mouse IL-12 and IL-15. Four independent experiments. Statistical analysis was performed using paired t-tests (Methods). i, In vivo radiance of LLC LeptoM cells delivered intracisternally into C57BL/6-Tyrc-2 mice overexpressing eGFP or Ifng in the leptomeninges treated with isotype or asialo-GM1 antibodies, 2 weeks after injection. j, In vivo radiance of LLC LeptoM cells delivered intracisternally into C57BL/6 Ifngr1−/− and Ifngr1+/+ mice treated with isotype or asialo-GM1 antibodies, 2 weeks after injection. k, In vivo radiance of LLC LeptoM cells delivered intracisternally into mice with WT NK cells and mice with ΔIfngr1 NK cells, 2 weeks after injection. l, NK products in the CSF of patients without and with LM, as determined using bead arrays. Statistical analysis was performed using multiple t-tests. m, In the leptomeninges, T cells produce IFNγ, supporting maturation of conventional DC2 cells into migratory CCR7+ DCs. These DCs produce IL-12 and IL-15 to support the survival and proliferation of NK cells, which control the expansion of metastatic cells.
We next examined communication between leptomeningeal CCR7+ DCs and NK cells. As determined by CITE-seq, mouse leptomeningeal CCR7+ DCs specifically produced IL-12 and IL-15, two cytokines that promote survival and proliferation of NK cells; leptomeningeal NK cells expressed their cognate receptors (Fig. 5d). Notably, production of IL-12 was significantly abrogated in Ifngr1-deficient CCR7+ DCs (Extended Data Fig. 13b). Mirroring findings in our mouse models, we detected increased levels of NK-cell-supporting cytokines in CSF from patients with LM (Fig. 5e), as well as transcripts of their receptors in human leptomeningeal NK cells (Fig. 5f). To examine this putative cell–cell communication, we cultured mouse splenic NK cells in human CSF obtained from patients with cancer with and without LM. CSF represents a notoriously nutrient-sparse environment with minimal growth factors38. Within LM-negative CSF, naive splenic NK cell survival was impaired compared to LM-positive CSF (Fig. 5g); this effect was rescued by the addition of recombinant mouse IL-12 and IL-15 (Fig. 5h).
To demonstrate the role of NK cells in IFNγ-dependent cancer control, we depleted NK cells using anti-asialo-GM1 antibodies in mice overexpressing Ifng in our AAV system (Fig. 3a). As expected, we observed control of tumour growth and extended survival in mice treated with control antibodies in the presence of leptomeningeal IFNγ (Fig. 5i and Extended Data Fig. 14a–e). This phenotype was abolished in mice with antibody-depleted NK cells, supporting a model whereby NK cells function as the leptomeningeal effector cells in the context of IFNγ. To determine whether this NK IFNγ response was direct or indirect, we next depleted NK cells in Ifngr1-deficient host mice. In this epistasis experiment, NK cell depletion in mice with non-functional IFNγ signalling did not further accelerate leptomeningeal cancer cell growth, confirming that IFNγ signalling precedes NK-cell-dependent cancer elimination (Fig. 5j). We further confirmed the dispensability of NK cell IFNγ signalling for leptomeningeal tumour control and NK cell proliferation in a host mouse with conditional Ifngr1 knockout in the NK cell population (Fig. 5k and Extended Data Fig. 14f). The impaired NK cell proliferation, observed in whole-body Ifngr1-deficient mice, was therefore not caused by direct impairment of IFNγ signalling in NK cells but, instead, indirectly through lack of IFNγ-induced cytokines (Extended Data Fig. 14g). We found evidence of NK cell activation in human LM in the form of moderately elevated levels of soluble FAS, granzyme A, perforin and granulysin, as well as enrichment of activated NK cells in the CSF of patients with LM (Fig. 5l and Supplementary Fig. 5d–f). Taken together, our data are consistent with a model in which T cell-derived leptomeningeal IFNγ acts on cDCs, supporting their maturation into CCR7+ DCs. These cells then produce a spectrum of lymphocyte-supporting cytokines, promoting NK cell proliferation and anti-leptomeningeal tumour action (Fig. 5m).
Discussion
We have defined the molecular interactions between metastatic cancer and immune cells within the leptomeninges. To capture this complex oncologic ecosystem, we used single-cell transcriptional and proteomic profiling of clinical samples. In doing so, we identified IFNγ as a key mediator of the anti-cancer response within the leptomeninges. To mechanistically dissect the growth suppressive action of leptomeningeal IFNγ, we generated several immunocompetent animal models of LM. Using an array of whole-body and conditional Ifng- and Ifngr1-deficient mouse lines, we found that, although leptomeningeal IFNγ attracts myeloid cells into the leptomeningeal space, it does not promote anti-tumour activity in the macrophage population. Rather, leptomeningeal IFNγ targets DCs, promoting cDC2 maturation. Notably, these DCs orchestrate anti-cancer activity in an antigen-independent manner, generating cytokine signals to support the cytotoxic action of natural killer cells—an anti-tumour response that is unique to the leptomeningeal space.
LM represents a fundamentally inflammatory pathology. Indeed, LM was originally described as carcinomatous meningitis, reflecting the characteristic abundant immune infiltrate and the purulent exudate found at autopsy39,40. Recent work demonstrates that certain aspects of leptomeningeal inflammation can support cancer cell growth: cancer cells within the leptomeningeal space respond to IL-8 and IL-6 to transcribe the iron-binding and iron-transport genes LCN2 and SLC22A176; cancer-cell-generated complement C3 disrupts the blood–CSF barrier to enrich the CSF and support cancer cell growth in the space3; and cancer cells use neuronal mimicry to trigger secretion of survival factors by meningeal macrophages41. However, inflammatory signalling does not universally support LM. Here we have identified leptomeningeal inflammatory signalling that can interrupt cancer cell growth through IFNγ. We identify elevation of leptomeningeal IFNγ as a hallmark of LM-induced pleocytosis across multiple tumour types. Moreover, higher CSF IFNγ at diagnosis portends a more favourable prognosis for these patients.
IFNγ is a classical tumour-suppressive cytokine, derived predominantly by T helper type 1 CD4+ T cells, CD8+ T cells, NK cells, NK T cells and minor immune cell populations42. Our investigation of IFNγ within the leptomeningeal space revealed anatomically distinct features: the proportion of immune cells expressing this protein, or its transcript, appeared to be insufficiently low even in the absence of malignancy, suggesting that the leptomeninges actively maintain low production of this pleiotropic cytokine to impede neurotoxicity9,20. IFNγ stimulates the recruitment of a wide variety of immune cell types into the tumour microenvironment, particularly through the upregulation of CXC chemokines CXCL9, CXCL10 and CXCL1142. The pleocytosis observed in LM can be to some extent explained by accumulation of these IFNγ-regulated chemokines. Both CC and CXC chemokines are substantially elevated in the leptomeninges of patients with systemic inflammation or prolonged COVID-19, yet their accumulation does not necessarily result in clinically relevant CSF pleocytosis, suggesting an additional level of control for immune cell entry into the CSF4,6,9,43. Why CSF IFNγ and its downstream ligands do not consistently result in leptomeningeal accumulation of immune cells remains an open question.
In immunocompetent settings, IFNγ prevents the establishment of spontaneous and chemically induced tumours by enhancing cancer cell recognition through increased antigen processing through MHC class I and II in the extracranial sites and brain44,45,46. In contrast to other anatomic compartments, the tumour-suppressive role of IFNγ within the leptomeninges was unexpectedly independent of both the adaptive immune system and monocytes and macrophages. Instead, leptomeningeal DCs represent the essential IFNγ target. Our systems-level approach suggests that metastasis renders the leptomeningeal space an unusually DC-rich environment, certainly when compared with extracranial sites. Indeed, cytometry analysis of STAT1 phosphorylation in the presence of LM was most apparent in DCs. Moreover, single-cell proteogenomic analysis of leptomeningeal DCs supported a role for IFNγ in cDC maturation into CCR7+ migratory DCs26. Trajectory analysis of mouse cDCs supports the assertion that these CCR7+ DCs are predominantly a product of cDC2 maturation. In extracranial tumours, both cDC1 and cDC2 cells equally contribute to the migratory DC pool33. In antigen-independent settings, these migratory DCs produce an array of immune cell pro-survival and proliferation factors. In the harsh leptomeningeal environment, these DC-generated signals are necessary to sustain effector cell viability and activation. Indeed, we show increased NK cell proliferation in the setting of Ifng overexpression, and that this is supported by the presence of migratory DC-derived signals including IL-12 and IL-15.
An improved understanding of LM-specific cancer-cell–immune-cell interactions suggests approaches to immuno-oncology within the CNS and prompts a more nuanced view of the immune system in the leptomeninges. Our findings demonstrate that leptomeningeal metastatic cancer cell growth is largely controlled by the innate immune system. This may explain modest outcomes in LM-focused clinical trials targeting adaptive immunity47,48,49. We propose that DC and NK cell-engaging therapies may act as more robust strategies to control this bleak complication of cancer50,51.
Methods
Human CSF
Patients with cancer undergoing routine clinical procedures including spinal tap, Ommaya reservoir tap or a ventricular shunt provided informed consent. CSF collected in excess of that needed for clinical care was reserved for this use under MSKCC Institutional Review Board-approved protocols 20-117, 18-505, 13-039, 12-245 and 06-107. Human CSF was processed as described previously6, de-identified and aliquoted. Cell-free CSF and CSF cell pellets were biobanked and stored at −80 °C until further analysis. Patient medical records and MRI scans were reviewed to confirm the LM status by neuro-oncologists (U.T.S., J.A.W. and A.B.), and clinical data necessary for this study were abstracted and de-identified by neuro-oncologists or clinical research assistants (R.E. and K.C.). Giemsa-stained cytospins were part of routine diagnostic assessment and were retrieved and reviewed by a neuropathologist (T.A.B.).
Human single-cell transcriptomics
Sample processing
Freshly collected CSF obtained by lumbar puncture was placed on ice and processed within 2 h, as described previously6. PBS-washed cells were encapsulated using the Chromium Single Cell 3’ Library and Gel Bead Kit V2 (10x Genomics) and sequenced on the NovaSeq 6000 system (Illumina). Raw and preprocessed data were deposited to the NCBI GEO under accession number GSE221522.
Data preprocessing, initial processing and batch correction
Raw FASTQC files were pre-processed with SEQC52,53 with human reference genome hg38, and dense SEQC matrices were imported into Python. Each sample was plotted as a histogram of total counts per cell barcode on the log scale, resulting in a distribution with multiple modes and the threshold to remove the smallest mode, containing empty droplets and low-quality cells, was defined manually. We next removed any genes that had counts equal to 0 after filtering. To remove doublets, we run the DoubletDetection method (parameters n_iter = 50, p_thres = 1e-7, voter_thres = 0.8)54. We outer joined the individual samples to retain all of the detected genes, filtered cells to a minimum count of UMI = 100 and minimum total expressed genes of 100. We initially detected 22,051 cells and retained 20,676 high-quality cells and 18,322 genes after filtering. We detected around 1,497 ± 898 genes per cell, about 6,268 ± 6,553 gene counts per cell, out of which 3.25 ± 2.99% were mitochondrial genes (values represent mean ± 1 s.d.). We normalized the library size, keeping the raw count matrix for downstream analyses and removed any genes expressed in fewer than five cells. For downstream analysis, we further removed mitochondrial genes (prefix MT-), ribosomal genes (prefix RPS- or RPL-) and haemoglobin genes (prefix HB-). We run Scanorama (default settings; knn = 20)55 on the resulting AnnData object to batch correct across patients. Batch correction was validated as follows: (1) cancer cells have higher interpatient heterogeneity (Supplementary Fig. 1), suggesting the absence of overcorrection; and (2) we identified and filtered out only few quasi-cancer cells from patients without LM after computational mixing (their presence was ruled out by pathologist during diagnostic cytology reading). This corrected matrix was used for visualization, but not for individual gene comparisons. We then run PCA (sc.pp.pca, n_components = 100). We constructed a k-nearest neighbour graph (k-NN) based on the 30 nearest neighbours and 100 principal components, using the Scanorama 100-dimensional matrix (instead of PCA matrix). We clustered the cells with Leiden clustering (resolution 2.0)56 and these Leiden clusters were merged according to major cell types, which were assigned on the basis of marker gene expression, as shown in Supplementary Fig. 1. The UMAP was computed with sc.tl.umap, using the default parameters. The interpatient heterogeneity was measured with Shannon entropy, Hj (ref. 57) (Supplementary Fig. 1g–i):
For each cell, the Shannon entropy measures the sample diversity of its nearest neighbours in the k-NN graph. Each sample was subsampled to contain 500 cells. If samples are well-mixed, the entropy of each cell will be high while, if samples are not well mixed, the entropies will tend to be low (this is true for cancer cells in general, which show extreme heterogeneity across patients). Quality-control plots are shown in Supplementary Fig. 1. Human LM+ single-cell transcriptomic data was retrieved from NCBI GEO GSE150660 (ref. 6). Raw and pre-processed data are available at the NCBI GEO under accession number GSE221522. All ten human samples were collected between December 2017 and May 2018 and were processed with the same pipeline.
Subsetting of cells for downstream analyses and visualization
Subsetting was performed by selecting cell clusters from major Leiden populations, shown in Fig. 1b. For analysis of DCs, the cDC and pDC clusters were subsetted and reclustered with sc.tl.umap and Leiden (resolution = 0.5). Cell type annotation was performed as follows: cDC1 cells are CLEC9A+XCR1+, cDC2 cells are CLEC10A+CD1C+, pDC cells are IRF7+TCF4+. Human LAMP3+ migratory DCs are LAMP3+CCR7+ (orthologous to mouse CCR7+ DCs; Supplementary Fig. 4). Two clusters bearing cDC2 signature were merged for further analyses. For analysis of NK, NK cluster was subsetted and reclustered with Leiden (resolution = 0.8), yielding in populations of cells with high SELL (CD62L) expression, further denoted as naive-like, and populations with low SELL expression, denoted as activated-like and characterized by the expression of CXCR6. For analysis of both cell types, we run Palantir with the default settings (n_components = 5, knn = 30), enabling us to access Markov affinity-based graph imputation of cells (MAGIC-imputed) cell counts, and these imputed cell counts were used only for visualization with 2D plots35,58. UMAP, t-SNE and heat-map plotting was performed using the Scanpy59 and scVelo60 toolkits. Embedding density was computed with sc.tl.embedding_density (Fig. 1c). For NK cell gene expression heat map, counts were first zero-centred with sc.pp.scale (Supplementary Fig. 5).
Gene signature analysis
Gene signatures were computed with the Scanpy function sc.tl.score_genes and projected onto the UMAP (Fig. 1b) and matrixplot. IFNγ gene signature 1 was created based on our mouse dataset, comparing the gene ranks in all cells from eGFP versus Ifng cancer-bearing mice to mimic the situation in our patients. Genes were ranked with sc.tl.rank_genes_groups, t-test method. This gene set contained the following genes: S100A9, S100A8, IRF7, PLAC8, ALOX5AP, FGL2, ISG15, CD74, PSAP, NAPSA, SLFN5, TALDO1, PLBD1, SPI1, MPEG1, LCN2, NAAA, TRIM30A, IRF8, NCF1, CBFA2T3, NGP, MS4A4C, CHIL3, CYBB, SLAMF7, RNASE6, RETNLG, CLEC12A, IFI205, CAMP, SLFN1, CKB, IGKC, IFIT3, AMICA1, LY86, OASL2, CSF2RA, CD24A, XCR1, OAS3, IFITM6, CLEC9A, IFIT2, LTF, WDFY4, LY6I, WFDC21 and USP18 (50 genes after removal of mouse-specific genes, including HLA). IFNγ gene signature 2 GSEA Hallmark interferon gamma response gene set was retrieved online (https://www.gsea-msigdb.org/gsea/msigdb/human/geneset/; 200 genes).
Human CSF targeted proteomics
The samples were processed and analysed essentially as described previously9. Biobanked CSF collected between 2015–2020 was aliquoted and stored at −80 °C at MSK Brain Tumor Center CSF Bank. Samples were slowly thawed on ice and 45 μl of CSF was mixed with 5 μl of 10% Triton X-100 (Sigma-Aldrich, T8787) in saline and incubated at room temperature for 2 h (final concentration of Triton X-100 was 1%). The samples were then dispensed in a randomized manner into 96-well PCR plates and stored at −80 °C until further analysis. Relative levels of proteins in two targeted panels were detected using proximity extension assay (Olink Target 96 Inflammation and Olink Target 96 Neuro Exploratory, Olink). As an additional control, LM− samples were retrieved from a previous study9 (CoV− cohort). Protein abundance values are shown in NPX units (on the log2 scale). The analytical range for each analyte is available online (www.olink.com).
Mouse strains and housing
All animal studies were approved by the MSKCC Institutional Animal Care and Use Committee under the protocol 18-01-002. WT C57BL/6 (JAX, 000664) were purchased from Jackson Laboratory or bred in-house. C57BL/6-Tyrc-2 (JAX, 000058, albino C57Bl/6) and BALB/c (JAX, 000651) animals were purchased from the Jackson Laboratory. NSG animals were obtained from MSKCC RARC Colony Management Group. Purchased mice were allowed to habituate for at least 1 week before manipulation and experimentation. Transgenic lines on the C57BL/6 background were purchased from the Jackson Laboratory and bred in house: Ifng-knockout line (B6.129S7-Ifngtm1Ts/J, JAX, 002287)15, Ifngr1-knockout line (B6.129S7-Ifngr1tm1Agt/J, JAX, 003288)16, Rag1 knockout line (B6.129S7-Rag1tm1Mom/J, JAX, 002216)61, double-reported knock-in/knockout Cx3cr1GFP/GFPCcr2RFP/RFP line (B6.129(Cg)-Cx3cr1tm1LittCcr2tm2.1Ifc/JernJ, JAX, 032127)62,63, congenic B6 CD45.1 line (B6.SJL-PtprcaPepcb/BoyJ, JAX, 002014), Zbtb46-DTR line (B6(Cg)-Zbtb46tm1(HBEGF)Mnz/J, JAX, 019506)27. For homozygous breeding, breeding pairs and randomly selected progenies used in the experiments were genotyped as recommended. For experiments that involved bioluminescence imaging where WT animals were not compared to transgenic lines, albino C57BL/6-Tyrc-2J animals were used. Mice in all experimental groups were age (±4 days), sex and fur-colour matched. Mice used in this study were housed under specific-pathogen-free conditions, in an environment with controlled temperature and humidity, under 12 h–12 h light–dark cycles (lights on and off at 06:00 and 18:00, respectively), and with access to regular chow and sterilized tap water ad libitum.
Compound transgenic mouse lines
We used the following alleles on C57BL/6 background to generate the compound strains: Clec9acre (C. Reis e Sousa)30, Ifngfl/fl (A. Rudensky), Ifngr1fl/fl (JAX, 025394)64, Lckcre (JAX, 003802)65, LysMcre (JAX, 004781)66, Nkp46cre (E. Vivier)67, Rosa26lsl-DTR (JAX, 007900)68, Rosa26lsl-eYFP (JAX, 007903)69, Xcr1cre-mCherry (JAX, 035435)70. To generate the Clec9acreRosa26lsl-DTR depletor line, heterozygous Clec9acreRosa26lsl-DTR mice (Brown laboratory) were crossed together. To generate the conditional NK cell Ifng-knockout line, heterozygous Nkp46creIfngwt/fl animals (Sun laboratory) were crossed to establish the Nkp46creIfngfl/fl colony. To generate the conditional T cell Ifng-knockout line T cellΔIfng, Nkp46creIfngwt/fl mice were crossed to Lckcre animals. The Nkp46cre allele was bred out and LckcreIfngfl/fl line was established. To generate the conditional myeloid cell Ifngr1-knockout line, LysMcre animals were crossed with Ifngr1fl/fl mice to establish the LysMcreIfngr1fl/fl line. To generate the conditional cDC Ifngr1-knockout line, heterozygous Clec9acreRosa26lsl-DTR animals (Brown laboratory) were crossed with Ifngr1fl/fl mice and the Rosa26lsl-DTR allele was bred out to establish the Clec9acreIfngr1fl/fl line. Similarly, to generate the conditional NK-cell Ifngr1-knockout line, heterozygous Nkp46creIfngwt/fl animals were crossed with Ifngr1fl/fl mice and the Ifngwt/fl allele was bred out to establish the Nkp46creIfngr1fl/fl line. Progeny was genotyped and the cre-negative littermates on homozygous flox/flox were used as controls. For DC lineage tracing, the homozygous Xcr1cre-mCherry line was crossed with the homozygous Rosa26lsl-eYFP line. Mice were housed as described above.
Cell culture
Mouse lung cancer LLC sublines were described previously3. Mouse breast cancer E0771 cells were a gift from E. E. Er. B16-F10 (CRL-6475), Yumm5.2 (CRL-3367), EMT6 (CRL-2755) and 4T1 cells (CRL-2539) were obtained from ATCC. LentiX 293T cells (632180) were obtained from Takara. PlasmoTest HEK Blue-2 cells (rep-pt1) were obtained from Invivogen. LLC, E0771 and B16 sublines and LentiX 293T and HEK Blue-2 cells were maintained in high-glucose DME (MSKCC Media Core), supplemented with 10% fetal bovine serum (FBS; Omega Scientific, FB-01) and 1% penicillin–streptomycin (Gibco, 15140163) or 1× Primocin (Invivogen, ant-pm-2). Yumm5.2 sublines were maintained in high-glucose DME:F12 (MSKCC Media Core), supplemented with 10% FBS, 1% non-essential amino acids (Gibco, 11140050) and 1% penicillin–streptomycin or 1× Primocin. 4T1 sublines were maintained in RPMI (MSKCC Media Core), supplemented with 10% FBS and 1% penicillin–streptomycin or 1× Primocin. EMT6 sublines were maintained in Waymouth’s (MSKCC Media Core), supplemented with 10% FBS and 1% penicillin–streptomycin or 1× Primocin. Cell lines were subcultured at least twice a week, replaced approximately after 6 weeks in culture with new stocks, stored in liquid nitrogen and routinely tested negative for mycoplasma contamination. Cell lines that were not obtained directly from ATCC (LLC, E0771) were authenticated with STR profiling (ATCC). Proliferation of vehicle-exposed or recombinant mouse IFNγ-exposed (BioLegend, 714006) cancer cells in vitro was measured with CellTiter-Glo luminescent cell viability assay (Promega, G7572) 72 h after seeding 500 cells per well into 96-well, white-walled plate (Corning).
Genetic engineering of mouse cancer cell lines
Plasmid DNA was amplified in NEB stable competent Escherichia coli (New England Biolabs, c3040i) or other E. coli strains provided by vendors, grown in LB broth (MSKCC Media Core) overnight and isolated with ZymoPURE II kit (Zymo Research, D4203). Mouse cancer cell lines generated in this study were engineered to constitutively express V5-tagged Firefly luciferase (pLenti-PGK-V5-Luc-Purow543-1, Addgene, 19360), gift from E. Campeau and P. Kaufman. Some LeptoM derivatives (LLC LeptoM, E0771 LeptoM, B16 LeptoM) used in the flow cytometry experiments were additionally engineered to constitutively express AmCyan fluorescent protein (pLV-EF1a-AmCyan1-IRES-Puro, Takara, 0039VCT). Lentiviral constructs for CRISPR–Cas9 editing in the pLV-hCas9:T2A:Bsd backbone were synthetized by VectorBuilder. sgRNA sequences expressed under the control of U6 promoter were as follows: sglacz, TGCGAATACGCCCACGCGAT; sgIfngr2.1, TGGACCTCCGAAAAACATCT; sgIfngr2.2, AGGGAACCTCACTTCCAAGT; sgIfngr2.3, TCTGTGATGTCCGTACAGTT. Lentiviral particles were prepared with LentiX 293T cell line using ecotropic, VSV-G pseudotyped lentiviral system and concentrator (Takara, 631276 and 631232), as recommended. Mouse cancer cell lines were spin-transduced (1,000g, 32 °C, 1 h) with concentrated lentiviral particles in complete culture medium containing 5 μg ml−1 hexadimethrine bromide (Santa Cruz, sc-134220) and selected for 5–7 days in complete medium containing 2–5 μg ml−1 puromycin (Gibco, A1113802) or 5–10 μg ml−1 blasticidin (Invivogen, ant-bl-1). CRISPR–Cas9 edited lines and control clones were single-cell sorted into 96-well plate. Gene function was assessed functionally (LLC, E0771 and B16 LeptoM; Extended Data Fig. 7), and DNA editing was confirmed with Sanger sequencing (LLC and E0771 LeptoM; not shown) after expansion.
Cancer cell injections
Cancer cells were injected into mice between 6 and 16 weeks of age. Mice were deeply anaesthetized in an insulated chamber perfused with 2–3% isoflurane (Covetrus, 11695067772) in medical air or with intraperitoneally delivered mixture of ketamine (100 mg per kg) and xylazine (10 mg per kg) in ultra-pure, sterile and pyrogen-free water for injection. Female mice were used for breast cancer models and both males and females in approximately 1:1 ratio for melanoma and lung cancer models, if not stated otherwise. Mice deceased within 72 h of injection were excluded from further analysis. Mouse hair was removed from the injection site, and the area was sterilized three times with ethanol. For intracisternal injection, 10 μl of cancer cell suspension in PBS was introduced into the cisterna magna using Hamilton syringe (Hamilton, HT80501) fitted with a 30 G needle, as described previously with minor modifications3. In brief, the mouse was positioned prone over a 15 ml conical tube to place cervical spine in flexion. The occiput was palpated, the needle was advanced 4 mm deep and the syringe content was slowly released into the cisterna magna. The syringe was then held in this position for another ten seconds and then carefully ejected to prevent the reflux of injected liquid. This procedure was tolerated well by the animals (success and survival rate > 95%). Mice displaying neurological symptoms after awakening were immediately euthanized. The number of cancer cells introduced intracisternally was as follows: 2,000 cells for LLC LeptoM; 4,000 cells for E0771 LeptoM; and 500 cells for B16 LeptoM, Yumm5.2 LeptoM, EMT6 LeptoM and 4T1 LeptoM cells. For intracardiac injections, 10,000 cells (for 4T1 or EMT6 sublines) or 50,000 cells (all other sublines) were injected in 50 μl saline using a 28-G insulin syringe into the left cardiac ventricle. For extracranial injections, cells were injected in 50 μl percutaneously into the fourth mammary fat pad (E0771, LeptoM; 500,000 cells), subcutaneously (LLC LeptoM; 200,000 cells) or intradermally (B16 LeptoM; 100,000 cells) using a 28-G insulin syringe. For primary tumour priming experiments (Extended Data Fig. 3), selected cell sublines were first injected into the extracranial sites. Then, 2 weeks later, mice were challenged with the same cancer cell sublines delivered intracisternally. Non-imaged tumours were shielded with a sheet of black watercolour paper to prevent capture of scattered photons. Images for cranium and extracranial site were therefore captured separately in the same mouse.
Quantification of tumour burden
The spread and growth of cancer cell lines engineered to express V5-tagged Firefly luciferase (lucV5) was monitored using non-invasive bioluminescence imaging (BLI). Mice were anaesthetized in an insulated chamber perfused with 2–3% isoflurane in medical air and injected retro-orbitally with 50 μl of sterile d-luciferin (15 mg ml−1, Goldbio, LUCK-5G) solution in PBS. BLI was captured using IVIS Spectrum-CT (Perkin Elmer). Data were recorded and processed using Living Image (v.4.7.2) software. Recorded images were quantified as cranial radiance. On the rare occasion when mice on the C57BL/6 background (without tyrosinase mutation) developed melanin spots preventing luciferase imaging, these animals were not included in the imaging analysis. For tumours in the mammary fat pad, intradermal and subcutaneous tumours were measured using calibrated digital callipers (VWR, 62379-531) or BLI. Tumour volumes are expressed as the product of the two largest diameters, as described previously71. Imaged areas were shaved to expose the skin before the imaging. Dark-furred mice that developed benign pigmented skin lesions in the shaved areas that interfere with BLI were not included in tumour quantification. Euthanasia was indicated when the tumour outgrowth from the injection site exceeded 1 cm3 or in the event of distress, including disease-specific averse neurological symptoms in the mouse such as head tilt, limb weakness or paralysis, and lethargy, whichever occurred first. Mice were monitored daily for the occurrence of neurological symptoms, bioluminescent imaging was generally performed once a week, palpable tumours were measured, and sizes were calculated twice a week. No animals exceeded the limits approved by the MSKCC IACUC.
Quantification of leptomeningeal tumour burden with image analysis
B16 melanoma sublines growing in 3D structures produce high amounts melanin that quenches light in a wide spectrum of wavelengths, interfering with accurate bioluminescent and fluorescence imaging. For these tumours, bioluminescence was therefore used solely to confirm the presence or identify the anatomical ___location of cancer. To overcome this limitation and to accurately quantify the tumour burden in the B16 LeptoM model, the brains from intracisternally injected mice were dissected, preserving the plaques of cancer, and fixed in formalin overnight. The brains were then carefully washed with tap water and placed into six-well dishes in 70% ethanol. Bright-field images of fixed brains (basilar plane) were taken using Lumar Stereoscope (Zeiss) against dark background. Data were processed with Fiji/ImageJ (v.2.0.0, NIH) as follows: images were converted to 8-bit, each brain was manually encircled and its area was recorded. The threshold for plaque measurement was first estimated in a small cohort to capture only the plaque areas and then applied to all subsequent measurements. The percentage of the area of cancer plaques covering the basilar surface of the brain was calculated as the area of plaques divided by the area of brain and multiplied by 100. As the 8-bit images were monochromatic, this method showed to be robust and reproducible throughout different measurements. Five control brains from mice without cancer, collected for different purposes, were measured and the area of darker structures above the pre-set threshold was less than 1% using this method.
Derivation of leptomeningeal and parenchymal metastatic cell lines
BrM cell lines (brain parenchyma-tropic)
A total of 50,000 parental cells was injected intracardially. Hematogenous dissemination was confirmed using BLI approximately 1 h after injection. After confirmation of brain colonization with BLI and development of late-stage cancer symptoms, mice were reinjected with luciferin and euthanized. The brains were dissected and imaged ex vivo to confirm colonization of parenchyma. Brains with overt lesions were minced, mechanically dissociated using GentleMACS (Miltenyi Biotec) and digested in a mixture of collagenase (100 U ml−1, Worthington, LS005273) and DNase I (10 U ml−1, Worthington, LS006333) in HG DME for 1 h at 37 °C, mechanically dissociated every 20 min. The suspension was then washed, filtered through a 70 μm mesh and seeded into corresponding complete culture medium, in which penicillin–streptomycin was replaced with Primocin. The medium was changed every day for 3 days, then every other day. Growing cancer cell colonies were expanded for three passages and named BrM1. These cells were then again injected intracardially and the whole procedure was repeated, leading to the establishment of BrM2 cell lines, competent to colonize brain parenchyma after hematogenous dissemination.
LeptoM cell lines (leptomeninges-tropic)
A total of 2,000 lucV5-expressing parental cancer cells in 10 μl saline was injected intracisternally. The presence in the CSF was confirmed with BLI approximately 1 h after injection. Mice were monitored weekly using BLI and daily checked for the presence of pathophysiological symptoms. When these mice developed neurological symptoms (moribund behaviour, head tilt, seizures, overall weakness) and cancer presence in the CSF was indicated by BLI, luciferin was injected retro-orbitally and mice were euthanized. The brain was dissected as described above and basilar sides of brains as well as basilar meninges of mouse were assessed with BLI post mortem. The cranial cavity and brain surface were then washed with approximately 3 ml of saline. This volume was collected, pelleted, resuspended in complete medium containing Primocin and maintained as described above for BrM cells. This procedure was repeated once for melanomas or three times for epithelial cancers, leading to the establishment of Inter cell lines. These Inter cells were then injected intracardially and the mice were monitored with BLI and treated as described above. Successfully expanded cancer cells that were isolated from these intracardially injected mice had the ability to colonize the leptomeninges and grow in the CSF, and were therefore named LeptoM cells. Three to five biologically independent sublines were successfully established per cell line. For transcriptomic analyses, these replicates were processed separately retaining the ID of founder mice. For further in vitro and in vivo manipulations, these replicates were pooled (at one-to-one ratios) and maintained under subconfluent conditions in vitro for a limited number of passages (less than 12).
RNA collection and extraction and transcriptomic analysis
Cancer cell lines were collected 24 h after initial seeding of approximately 1 × 106 cells per 100 mm plate by direct lysis with RLT buffer (Qiagen, component of RNeasy kits). RNA from cell lines was isolated with RNeasy Plus Mini Kit (Qiagen, 74136), and sequenced and analysed as described previously72. Resulting HTSeq73 matrices from bulk transcriptome were processed in R Studio with DESeq274. Data from LLC cell lines were retrieved from NCBI GEO GSE83132. Newly generated raw and preprocessed data are available through NCBI GEO under accession number GSE221358.
Collection of mouse CSF and leptomeningeal immune cells
Mice were deeply anaesthetized using ketamine–xylazine and transcardially perfused with sterile, ice-cold PBS. Mice were positioned as described in the ‘Cancer cell injections’ section, and CSF was collected through the cisternal puncture into a PBS-flushed syringe fitted with a 30-G needle. Approximately 5–15 μl of CSF was collected from each single mouse using this procedure. Blood-contaminated samples were discarded. CSF was flash-frozen on dry ice and stored at −80 °C until analysis, or diluted in 200 μl of 4% methanol-free paraformaldehyde (Electron Microscopy Sciences, 15714-S) and spun onto microscopy slides to produce cytospins. These were then left to air dry and stained with haematoxylin QS (Vector Biolabs, H-3404-100). Leptomeningeal immune cells were collected as described previously6 and processed further for downstream applications, as described in the corresponding sections. Controls were prepared from mechanically dissociated spleens (GentleMACS, spleen program) and enzymatically dissociated brains and lungs (dissociation described above, mechanical assistance was performed with GentleMACS, brain and lung programs).
Intracisternal delivery of recombinant proteins and AAV particles
Vehicle (PBS) or a 10 ng or 25 ng dose of recombinant mouse IFNγ (BioLegend, 714006) in total volume of 10 μl was initially delivered with cancer cell injection, followed by weekly administration, as described above. Heat-inactivated IFNγ was prepared by incubating vehicle or vehicle-diluted IFNγ at 95 °C for 15 min and allowed to cool on ice before administration. Mouse Ifng (NM_008337.4) or eGFP sequences were inserted into AAV expression vector (pscAAV backbone under the control of CMV promoter) and used for packaging into AAV5 particles that were ultrapurified for in vivo applications (VectorBuilder). Genomic content was estimated with PCR. A total of 5 μl of vehicle-diluted AAV5 suspension (1 × 1013 genome copies per ml) was slowly infused into mouse leptomeninges intracisternally and mice were allowed to rest for at least 2 weeks before further manipulation.
Mouse single-cell proteogenomics
Sample processing
Cx3cr1GFP/GFPCcr2RFP/RFP mice were crossed with WT C56BL/6 mice and the resulting female and male Cx3cr1+/GFPCcr2+/RFP progeny were intracisternally infused with AAV and LLC LeptoM cancer cells, as described above and in Supplementary Fig. 3. Leptomeningeal cells from six animals per group were isolated and resuspended in Cell Staining Buffer (BioLegend, 420201). In total, we profiled leptomeningeal immune cells from 24 mice and 4 different conditions. To limit non-specific antibody binding, cells from each mouse were incubated with TruStain FcX (BioLegend, 101320), subsequently barcoded with TotalSeq-A anti-mouse hashtags 1 to 6 (BioLegend), listed in Supplementary Table 4, and washed. Cells from these six mice were then pooled, resulting in four independent samples, and stained with a custom TotalSeq-A panel (BioLegend), consisting of 198 antibodies targeting cell-surface epitopes and non-targeting isotype controls, listed in table Supplementary Table 5, to facilitate identification and origin of selected immune cell types (such as in Fig. 4). Dead cells and debris were removed with LeviCell (LevitasBio), and washed cells were counted, encapsulated using the Chromium Single Cell 3’ GEM Library and Gel Bead Kit V3.1 (10x Genomics) and sequenced on the NovaSeq 6000 system. Quality control plots are shown in Supplementary Fig. 3. Raw and preprocessed data are available through NCBI GEO under accession number GSE221593.
Data preprocessing and initial processing
Raw FASTQC files were preprocessed with SEQC52,53 with modified mouse reference genome mm10 that included GFP, RFP and AmCyan sequences, and preprocessed as human samples, with the exception that no batch correction was applied. Each sample was processed separately. Cell filtering and doublet removal with DoubletDetection (p_thresh=1e-16, voter_thresh=0.5, n_iters=25, use_phenograph=False, standard_scaling=True)54 was performed as described above for human samples, we initially detected 54,781 cells and 20,804 genes and retained 46,852 high-quality cells and 18,277 genes after filtering out low-quality cells and non-immune cell populations. We detected around 1,387 ± 866 genes per cell, about 4,374 ± 5,483 gene counts per cell, out of which 3.15 ± 2.62% were mitochondrial genes (values represent mean ± 1 s.d.). The Shannon entropy for this uncorrected mouse dataset was computed as described above for human data. AnnData files for each sample were then merged after filtering and doublet removal by an outer join. Erythrocyte genes (HBA-A1, HBB-BT, HBA-A2, HBB-BS, ALAS2, HBB-BT, HP and BPGM) and CD41 protein signal (platelet marker) were filtered out, in an addition to mitochondrial (prefix MT-) and ribosomal genes (prefix RPS- or RPL-). HTO and CITE-seq data were demultiplexed with cite-seq-count75, using the default parameters applied on the whitelist of cells that passed the filtering step based on RNA quality, as described above. RNA and protein data (HTO and CITE) were integrated with totalVI, facilitating identification of immune cell subtypes using both gene and surface protein expression (default settings with top 4,000 highly variable genes (HVGs))76. HTOs were assigned based on maximum number of observed counts (Supplementary Fig. 3e). UMAP k-NN graph and Leiden clustering56 in this dataset was computed using sc.pp.neighbours59 and totalVI processed latent variables. Leiden clusters were merged according to major cell types, which were assigned based on marker gene and surface protein expression, as showed in Supplementary Fig. 2 (HVGs).
Subsetting of cells for downstream analyses, plotting and visualization
Plotting was performed using Scanpy (UMAP, t-SNE, heat maps)59 and scVelo (UMAP, t-SNE; this package was not used to infer RNA velocity)60. Embedding density was computed with sc.tl.embedding_density (Fig. 2c). Cell cycle prediction was adapted from tl.score_genes_cell_cycle60 (Fig. 4j,k and Fig. 5c). Subsetting was performed by selecting cell clusters from major populations, shown in Fig. 2b. We included cells from all four conditions, shown in Supplementary Fig. 3: cells isolated from naive, vehicle-injected or LLC LeptoM-injected animals that were overexpressing eGFP (control gene) or Ifng specifically in the leptomeninges. For analysis of DCs, cDC and pDC clusters were subsetted, these cells were expressing CD11c (pan-DC marker) on cell surface. For analysis of NK cells, NK and proliferative T/NK clusters were subsetted to ensure proper representation of all NK cells. These cells were reclustered with Leiden (resolution = 0.7), and clusters expressing CD3 and TCRβ cell surface markers were excluded, retaining only bona fide NK cells, characterized as Nk1.1+CD3−TCRβ−. For analysis of both cell types, we run Palantir (default settings, n_components = 5, knn = 30) that allowed us to (1) compute diffusion components, used for t-SNE re-embeddings; and (2) access MAGIC-imputed cell counts35,58 (Figs. 4d and 5a,d). These imputed cell counts were used only for visualization with 2D plots. t-SNE plots were refitted using multiscale coordinates that are based on diffusion components obtained with Palantir (n_components = 5, knn = 30). Subsetted DCs were refitted onto t-SNE using Palantir multiscale coordinates and annotated with initial Leiden loadings to identify four typical DC populations. We considered both gene expression data (shown as a heat map in Supplementary Fig. 4a) and cell surface signals: cDC1 cells are XCR1+, cDC2 cells are CD11b+, pDC cells are B220+ and CCR7+ cells express CCR7 (Fig. 4e and Extended Data Fig. 12d). Subsetted NK cells were refitted onto t-SNE using Palantir multiscale coordinates and reclustered with Leiden (resolution = 0.3), resulting in the identification of four putative cell states. Naive NK cells expressed high cell surface levels of CD62L (encoded by SELL), while activated and proliferative cells had low CD62L levels. Proliferative cells also expressed genes associated with cell cycling, such as MKI67, TOP2A and HMGB2. Senescent cells expressed CD55 and KLGR1 on their cell surface (Fig. 5b). Cancer cells, characterized by the expression of keratin genes and CD63, were subsetted as a cancer cluster and were visualized with UMAP without re-embedding. Cancer cell gene signatures were computed with GSEApy (Extended Data Fig. 13j,k; cut-offs are provided in the corresponding figure legends) (https://github.com/zqfang/GSEApy).
Trajectory analysis
To predict the maturation trajectories of cDCs in normal, non-perturbed steady-state mouse leptomeninges and leptomeninges with metastasis, we subsetted CD11c+ cDCs from naive and cancer-bearing mice overexpressing eGFP only (cDC cluster and eGFP condition). We first used CellRank to identify putative trajectories without the need for initial or terminal state selection34. We filtered out genes present in less than ten cells, normalized counts per cell and with log(X + 1) and extracted HVGs with Scanpy’s functions sc.pp.filter_genes, sc.pp.normalize_total, sc.pp.log1p and sc.pp.highly_variable_genes. We retained 2,635 cells and 2,090 cDC-expressed HVGs. We recomputed PCA with sc.pp.pca (n_comps = 50, zero_centered = True) and refitted the t-SNE plot with the top nine diffusion components in multiscale space (n_components=9, knn=15); this t-SNE map was used for further visualization. We used cytoTRACE kernel77, which enabled us to assess plausible and biologically traceable cell transitions, following their trajectory from more primitive to mature cells. We imputed gene counts from normalized and filtered count matrix with scv.pp.moments with the default parameters (n_pcs = 30, n_neighbors = 30) and initialized CellRank’s cytoTRACEkernel with the default parameters. Transition matrix was computed (threshold_scheme = hard). Given that this approach provides qualitative insights into the transition matrix by iteratively choosing the next cell based on the current cell’s transition probabilities, we further compared two additional settings: (1) we did not specify from which cells or condition to select starting point (start_ixs = None); or (2) we selected all cells from naive eGFP-overexpressing mouse as the starting points. Both approaches identified CCR7+ DCs as mature end points, and to remain agnostic to the initiation, we continued the analysis without initial cells or states being defined (n_sim = 100). We used GPCCA estimator (generalized Perron cluster analysis)78 to coarse-grain a discrete Markov chain into a set of macrostates and compute coarse-grained transition probabilities among the macrostates. We identified three macrostates and assigned each cell their dominant microstate membership. These results suggested that the cDC2 population is prone to maturate towards CCR7+ DCs, with an insignificant contribution of the cDC1 population (Extended Data Fig. 12f–h). CellRank prediction was corroborated by an analysis using Palantir (n_components = 9, knn = 15, num_waypoints = 500)35 that identified the cDC2 population as the one with the highest entropy (maturation potential), and this observation was robust to changes in the number of diffusion components, neighbours or waypoints (Extended Data Fig. 12i,j). We dissected the cDC2-to-CCR7 DC transition axis and plotted smoothened gene trends along the predicted Palantir pseudotime axis (Fig. 4f).
Bone marrow chimeras
For DTR depletion experiments, male C57BL/6-Tyrc-2 mice were initially anaesthetized with 2–3% isoflurane in medical air and restrained in ventilated conical plastic tubes. Animals were placed in a prone position and irradiated using X-RAD320 irradiator (Precision) with the following settings: 250 kV; 12 mA; using 0.25 mm copper filter; distance of radiation source to the animal body, 50 cm; irradiation field, 20 × 20 cm; dose rate, 117.5 cGy min−1. Five mice were fitted into the radiation field and received and two cycles of 5.5 Gy total body radiation 6 h apart. Immediately after completion of the irradiation procedure, animals were returned to their cages and fed with sulfatrim-enriched diet for the duration of this experiment. Within 24 h, mice were retro-orbitally infused with approximately 1 × 107 bone marrow cells from multiple pooled WT or Zbtb46-DTR+/+ C57BL/6 donors. Bone marrow cells were sterilely isolated from femur and tibia. Inner bone marrow was exposed and placed inside a 0.6 ml PCR tube with small hole punched in the bottom. The PCR tube was placed in 1.5 ml microcentrifuge tube and the samples were centrifuged to collect and pellet the bone marrow cells. Cells were counted and resuspended in sterile PBS. For head-shielded chimeras, female CD45.2+ C57BL/6-Tyrc-2 mice were initially anaesthetized with ketamine–xylazine. Mice were placed into the irradiator with their head positioned under a custom-made, 5 mm thick lead frame. Three mice were fitted into this field and received a single dose of 9 Gy extracranial radiation. Mice were left to recover from anaesthesia and returned to their cages. Mice fed with sulfatrim-enriched diet for the first 3 months after irradiation. Within 24 h, mice were retro-orbitally infused with approximately 2 × 107 bone marrow cells from multiple pooled congenic CD45.1+ female donors (JAX, 002014), isolated as described above. Mice were left to recover after the transplant for 4 months before further experimentation. The recombination efficiency (CD45.1-to-CD45.2 ratio) was quantified at the end point in cells isolated from femoral and cranial bone marrow as described previously79, and in heparinized blood collected by cardiac puncture. Leptomeningeal cells were used for immunophenotyping with flow cytometry to assess the cranial and extracranial origin of selected immune cell types.
Immune cell depletions
cDC progenitors were depleted in bone marrow chimeras that received WT or Zbtb46-DTR+/+ donor cells with DTx (Sigma-Aldrich, D0564), diluted in PBS and delivered intraperitoneally. The initial dose of 400 ng DTx was injected 1 day before cancer cell implantation, followed by bi-weekly injection of 100 ng. Both WT and Zbtb46-DTR+/+ cohorts were receiving DTx. In experiments with only Zbtb46-DTR+/+ chimeras, approximately half of the mice received PBS as a vehicle. cDC progenitors were depleted in Clec9acreRosa26lsl-DTR mice, Clec9acre littermates (DTR genotype negative) were used as controls. DTx dosing was the same as for Zbtb46-DTR chimeras. NK cells were depleted with polyclonal rabbit anti-mouse asialo GM1 (Poly21460; BioLegend, 146002), rabbit polyclonal IgG was used as control (Invitrogen, 02-610-2). Both antibodies were reconstituted with PBS. An initial dose of 50 μg was instilled 1 day before cancer cell implantation, followed by bi-weekly injections of 50 μg until the end point (see the schematic in Fig. 5i). T cells were inactivated and depleted with anti-mouse CD3ε (145-2C11, BE0001, BioXCell), Armenian hamster IgG was used as control (BE0091, BioXCell). Anti-CD3ε antibodies were diluted in pH 7.0 buffer (IP0070, BioXCell). Isotype was co-diluted with rabbit polyclonal IgG for the experiment with parallel depletion of NK cells and T cells (Extended Data Fig. 4). An initial dose of 100 μg was instilled 1 day before cancer cell implantation, followed by tri-weekly injections of 100 μg until the end point.
Flow cytometry
Single-cell suspensions were prepared as described above and previously6. After filtering through a 70-μm filter and washing with 2 mM EDTA and 1% BSA in PBS, non-specific binding sites were blocked with TruStain FcX (BioLegend, 101320) diluted in PBS, supplemented with 10% rat serum (Sigma-Aldrich, R9759) for 10 min on ice. Antibodies against surface antigens were diluted in reconstituted Brilliant Stain Buffer Plus (BD, 566385), supplemented with 5% rat serum. Surface antigens were stained for 15 min on ice, except for panels that included CCR7 staining and were stained for 20 min at ambient temperature. LIVE/DEAD Green/Violet/FarRed Dead Cell Stain kits (Life Technologies, L34969, L34963, L34973, respectively), DAPI (Molecular Probes, D1306) or propidium iodide (Thermo Fisher Scientific, P3566) were used as viability stains. Buffer without BSA was used before LIVE/DEAD staining, which was performed for 15 min on ice. Red blood cells were lysed with 1× ACK buffer or 1× eBioscience RBC lysis buffer (Invitrogen, 00-4300-54) for 5 min at ambient temperature.
For cytokine production analysis, leptomeningeal isolates were resuspended in serum-free IMDM and incubated (MSKCC Media Core) with or without addition of brefeldin A (BioLegend, 420601), ionomycin (StemCell Technologies, 73722) and phorbol 12-myristate 13-acetate (PMA; Invivogen, tlrl-pma), for 2 h at 37 °C. Where the intracellular staining was performed, cells were further fixed with IC fixation buffer for 20 min (Invitrogen, 00-8222-49) at room temperature, permeabilized and stained with antibodies against intracellular markers in 1× permeabilization buffer for 1 h (Invitrogen, 00-8333-56) and analysed. For pSTAT1 transcription factor staining, cells were processed using True-Nuclear Buffer Set (BioLegend, 424401) or FOXP3 Fix/Perm buffer set (BioLegend, 421403) and analysed. MHC class I levels of vehicle- or recombinant mouse IFNγ-exposed (BioLegend, 714006) cells in vitro was measured 24 h after treatment. Data were recorded using LSR Fortessa (BD). Gating and analysis were performed essentially as described previously6,80, using unstained samples, isotype-stained samples and/or FMO controls (Supplementary Fig. 6). During the analysis of ex vivo profilings, B cells were assigned as B220+, T cells as CD3+, monocytes-macrophages as CD11b and Ly6C+ or F4/80+, neutrophils as Ly6G+, NK cells as Nk1.1+, conventional DCs as CD11c+MHC-II+ (B220−) and pDCs as CD11c+MHC-II+B220+. The cDC1 population was gated as XCR1+ cDCs, and the cDC2 population was gated as CD11b+ cDCs. Typically, percentages of all leukocytes (CD45+) assigned with high confidence are plotted. A list of the antibodies used for flow cytometry is provided in Supplementary Table 6.
Mature CCR7+ DC lineage tracing
For cell surface protein mapping, we used C57BL/6-Tyrc-2 mice. For genetic lineage tracing, we used the Xcr1cre-mCherry mouse strain (JAX, 035435) crossed with Rosa26lsl-eYFP (JAX, 007903). The expression of XCR1-driven mCherry was hemizygous. Cell suspensions from leptomeninges and spleen were stained with a DC-focused antibody panel (CD45 BUV395, CD11c BUV737, MHC-II IA/IE PerCP, XCR1 BV650, CD11b PE/Cy7, CCR7 APC, B202 AlexaFluor 700; Supplementary Table 6) and dead cells were marked with DAPI. Live, single-cell CD45+ leukocytes were selected and pDCs were excluded based on B220 expression, conventional DCs were then gated as CD11c+MHC-II+ cells. These were further divided into XCR1+ cDC1 and CD11b+ cDC2 subtypes. Expression of CCR7+ was established based on the FMO control. XCR1 and CD11b cell surface protein expression in cDCs is typically mutually exclusive. Mature CCR7+ DCs of cDC1 origin therefore co-expressed CCR7 and XCR1, whereas mature CCR7+ DCs of cDC2 origin co-expressed CCR7 and CD11b. XCR1+ cDC1 cells, both immature and mature, expressed mCherry and mCherry expression patterns reflecting the antibody staining. Mature CCR7+ DCs of cDC1 origin in this experiment therefore co-expressed XCR1, mCherry and CCR7. We included a chronic systemic inflammation control by intraperitoneal LPS delivery for 2 weeks (6 μg dose in saline, three times a week, E. coli O55:B5 lipopolysaccharide, Sigma-Aldrich, L2880)6. Staining and analysis was performed as described above.
Soluble protein detection in plasma and CSF
Solute analytes in the human and mouse CSF were analysed using following multiplexed bead arrays, used as recommended by the manufacturer: LEGENDPlex mouse anti-virus response (BioLegend, 740622), LEGENDPlex human CD8/NK panel (BioLegend, 740267). IFNγ levels in human CSF were measured with human IFNγ Quantikine HS ELISA Kit (R&D, HSDIF0).
NK cell in vitro survival assay
NK cells were enriched from dissociated spleens of female and male C57BL/6 mice with MojoSort mouse NK cell isolation kit (BioLegend, 480049). Approximately 20,000 cells were seeded into a 1:1 mixture of HG DME and human CSF from patients with cancer without or with LM containing 10 ng ml−1 recombinant human IL-2 (BioLegend, 589102), into a 96-well plate. Cells were incubated for 24 h with or without the addition of 1 ng ml−1 or recombinant mouse IL-12p70 (BioLegend, 577002) and recombinant mouse IL-15 (PeproTech, 210-15-10ug). Viability and cell counts were assessed with cytometry.
NK cell in vivo proliferation
Selected mice with injected with a dose of 100 μg 5-ethynyl-2′-deoxyuridine (EdU) in 100 μl saline intraperitoneally 4 h before transcardial perfusion with saline. Leptomeningeal cells were then collected and processed for flow cytometry. Surface staining was performed with polymer dye-tagged antibodies (CD45 BUV396 and Nk1.1 BV650; Supplementary Table 6), cells were stained for viability with LIVE/DEAD FarRed dye and further processed as described above. Cells were fixed with 1% MeOH-free paraformaldehyde in PBS and EdU click-it reaction was performed with Click-iT EdU AlexaFluor488 Flow Cytometry Assay Kit as recommended (C10632, Thermo Fisher Scientific).
Histology
Tissue from euthanized mice was fixed in 10% formalin overnight, thoroughly washed in tap water, sliced and stored in 70% ethanol until being embedded in paraffin. Paraffin-embedded blocks were then cut into 5-μm-thick sections and placed onto microscopy slides. H&E staining was performed by the MSKCC Molecular Cytology Core. Myelin staining was performed using the Luxol Fast Blue stain kit (Abcam, ab150675). Immunofluorescence was performed as described previously6, using following primary antibodies: CD11c (hamster, 1:50, Novus, NBP1-06651 and NB110-97871, used in combination), cleaved caspase 3 (rabbit, 1:200, Cell Signaling Technology, 9661S, CNPase (mouse, 1:1000, Abcam, ab6319), DCX (sheep, 1:200, R&D, AF10025), GFAP (goat, 1:500, Abcam, ab53554), IBA1 (rabbit, 1:500, Invitrogen, PA5-27436; and goat, 1:500, Novus, NB100-1028), MBP (mouse, 1:100, R&D, MAB42282), NeuN (mouse, 1:100–1:500, Sigma-Aldrich, MAB377) and OLIG2 (goat, 1:200, R&D, AF2418). AF488-, Cy3- and AF647-conjugated, anti-mouse, goat, rabbit and sheep secondary antibodies were obtained from Jackson ImmunoResearch; AF647-conjugated anti-hamster secondary antibodies were obtained from Abcam. For antibodies of murine origin applied on mouse tissue, the endogenous IgG was first blocked with reconstituted VisUBlock Mouse (R&D, VB001-01ML). DAPI (Molecular Probes, D1306) was used as nuclear counterstain. Autofluorescence was quenched with Vector TrueView (Vector Laboratories, sp-8400). Slides were scanned with a Mirax slide scanner (Zeiss), and images for further analysis were exported with CaseViewer (3DHISTECH).
Quantification of immunofluorescence imaging
Quantification of IBA1+ myeloid cells in the choroid plexus and IBA1+ microglia in cortical layers 3 and 4 was performed essentially as described previously19. Cleaved-caspase-3-positive cells in cancer plaques and clusters in leptomeninges were counted manually in fields of view of approximately equal size. The thickness of the periventricular GFAP+ layer was measured in both hemispheres and averaged per mouse. For MBP, the area of MBP+ tracts in both hemispheres was quantified and averaged per mouse. The relative fluorescence of DCX progenitors in the hippocampus was analysed in the corresponding AlexaFluor647 channel and reported as RFU. Cancer plaques in timepoint-matched AVV5-Ifng animals are rare; 2–3 fields of view per brain were extracted and the exact animal sample size and number of fields of view are stated in the corresponding images. Analysis of NeuN was done in the motor and somatosensory cortex in two regions. Region 1 covers layers 1–4 and region 2 covers layer 5 and 6. One section per animal was analysed and the chosen sections were between levels −0.18 mm to −0.196 mm relative to bregma. OLIG2 was quantified in the corpus callosum, spanning a lateral area from 0–1.7 mm relative to bregma; and together with CNPase in also in subcortical and cortical region above the corpus callosum. All image analyses were performed in Fiji/ImageJ81.
Statistical analysis and reproducibility
Plotting and statistical analysis were performed using Prism v.8.1.0 (GraphPad Software). Typically, experiments performed in vivo and ex vivo were compared using Mann–Whitney U-tests, unless specified otherwise. Metastasis incidences in the Ifngr1-proficient and -deficient mice were analysed using a χ2 test (Extended Data Fig. 6). Phenotypic changes in the in vitro cultured cancer cells after exposure to IFNγ were analysed using a t-test (Extended Data Figs. 7 and 8). Flow cytometry profiling of immune cell proportions across various conditions was analysed with multiple Mann–Whitney t-tests with Holm–Šídák correction. In the box plots (box and whisker plots), the box limits extend from the 25th to 75th percentile and the whiskers show minimum to maximum values. The results from single-cell analyses were plotted in Python. Bulk RNA-seq data were processed in R Studio. Mouse experiments are typically from at least two independent repetitions, except for Figs. 3g and 4a and Extended Data Figs. 3a,b, 4f–i, 5c,d, 7g,h, 8j, 11a,j and 14c–f; these in vivo experiments were performed once. Whenever possible, mice were randomly allocated into treatment groups. This was not possible in experiments with transgenic animals. Investigators were not blinded to the genotype or treatment over the course of experiment. Sample size and exact P values are included in the figures. Sample sizes were not predetermined. Critical mouse experiments (Figs. 2 and 3 and associated data) were reproduced by two independent investigators. Animal exclusion criteria for animal experiments (death within 3 days of injection or appearance of pigmented spots that interfered with BLI) are described above. One human CSF sample was excluded from IFNγ ELISA analysis as clinical chart review revealed the presence of sterile meningitis in this individual at the time of CSF collection.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
RNA-seq datasets were deposited online in the NCBI Gene Expression Omnibus (GEO) under the accession numbers GSE221358 (bulk RNA-seq), GSE221593 (mouse single-cell proteogenomics) and GSE221522 (human scRNA-seq). Commercially available materials can be obtained from vendors. Renewable materials generated in this study are available from the corresponding author after signing the MSKCC material transfer agreement. Human samples used in this study are a limited biological resource, and are not available for further distribution. Source data are provided with this paper.
References
Wilcox, J. A. et al. Leptomeningeal metastases from solid tumors: a SNO and ASCO consensus review on clinical management and future directions. Neuro Oncol. https://doi.org/10.1093/neuonc/noae103 (2024).
Klemm, F. et al. Interrogation of the microenvironmental landscape in brain tumors reveals disease-specific alterations of immune cells. Cell 181, 1643–1660 (2020).
Boire, A. et al. Complement component 3 adapts the cerebrospinal fluid for leptomeningeal metastasis. Cell 168, 1101–1113 (2017).
Smalley, I. et al. Proteomic analysis of CSF from patients with leptomeningeal melanoma metastases identifies signatures associated with disease progression and therapeutic resistance. Clin. Cancer Res. 26, 2163–2175 (2020).
Yoo, B. C. et al. Cerebrospinal fluid metabolomic profiles can discriminate patients with leptomeningeal carcinomatosis from patients at high risk for leptomeningeal metastasis. Oncotarget 8, 101203–101214 (2017).
Chi, Y. et al. Cancer cells deploy lipocalin-2 to collect limiting iron in leptomeningeal metastasis. Science 369, 276–282 (2020).
Smalley, I. et al. Single-cell characterization of the immune microenvironment of melanoma brain and leptomeningeal metastases. Clin. Cancer Res. 27, 4109–4125 (2021).
Prakadan, S. M. et al. Genomic and transcriptomic correlates of immunotherapy response within the tumor microenvironment of leptomeningeal metastases. Nat. Commun. 12, 5955 (2021).
Remsik, J. et al. Inflammatory leptomeningeal cytokines mediate COVID-19 neurologic symptoms in cancer patients. Cancer Cell 39, 276–283 (2021).
Law, V. et al. A preclinical model of patient-derived cerebrospinal fluid circulating tumor cells for experimental therapeutics in leptomeningeal disease from melanoma. Neuro Oncol. https://doi.org/10.1093/neuonc/noac054 (2022).
Wang, N., Bertalan, M. S. & Brastianos, P. K. Leptomeningeal metastasis from systemic cancer: review and update on management. Cancer 124, 21–35 (2018).
Stoeckius, M. et al. Simultaneous epitope and transcriptome measurement in single cells. Nat. Methods 14, 865–868 (2017).
Eshleman, E. M., Bortell, N., McDermott, D. S., Crisler, W. J. & Lenz, L. L. Myeloid cell responsiveness to interferon-gamma is sufficient for initial resistance to Listeria monocytogenes. Curr. Res. Immunol. 1, 1–9 (2020).
Ivashkiv, L. B. IFNγ: signalling, epigenetics and roles in immunity, metabolism, disease and cancer immunotherapy. Nat. Rev. Immunol. 18, 545–558 (2018).
Dalton, D. K. et al. Multiple defects of immune cell function in mice with disrupted interferon-γ genes. Science 259, 1739–1742 (1993).
Huang, S. et al. Immune response in mice that lack the interferon-gamma receptor. Science 259, 1742–1745 (1993).
Castro, F., Cardoso, A. P., Goncalves, R. M., Serre, K. & Oliveira, M. J. Interferon-gamma at the crossroads of tumor immune surveillance or evasion. Front. Immunol. 9, 847 (2018).
Moris, G. & Garcia-Monco, J. C. The challenge of drug-induced aseptic meningitis revisited. JAMA Intern. Med. 174, 1511–1512 (2014).
Cui, J. et al. Inflammation of the embryonic choroid plexus barrier following maternal immune activation. Dev. Cell 55, 617–628 (2020).
Herrlinger, U., Weller, M. & Schabet, M. New aspects of immunotherapy of leptomeningeal metastasis. J. Neurooncol. 38, 233–239 (1998).
Kaya, T. et al. CD8+ T cells induce interferon-responsive oligodendrocytes and microglia in white matter aging. Nat. Neurosci. 25, 1446–1457 (2022).
Wang, Y. et al. STAT1/IRF-1 signaling pathway mediates the injurious effect of interferon-gamma on oligodendrocyte progenitor cells. Glia 58, 195–208 (2010).
Baruch, K. et al. CNS-specific immunity at the choroid plexus shifts toward destructive Th2 inflammation in brain aging. Proc. Natl Acad. Sci. USA 110, 2264–2269 (2013).
Shechter, R. et al. Recruitment of beneficial M2 macrophages to injured spinal cord is orchestrated by remote brain choroid plexus. Immunity 38, 555–569 (2013).
Abram, C. L., Roberge, G. L., Hu, Y. & Lowell, C. A. Comparative analysis of the efficiency and specificity of myeloid-Cre deleting strains using ROSA-EYFP reporter mice. J. Immunol. Methods 408, 89–100 (2014).
Nirschl, C. J. et al. IFNγ-dependent tissue-immune homeostasis is co-opted in the tumor microenvironment. Cell 170, 127–141 (2017).
Meredith, M. M. et al. Expression of the zinc finger transcription factor zDC (Zbtb46, Btbd4) defines the classical dendritic cell lineage. J. Exp. Med. 209, 1153–1165 (2012).
Akagbosu, B. et al. Novel antigen-presenting cell imparts Treg-dependent tolerance to gut microbiota. Nature 610, 752–760 (2022).
Zhou, W. et al. ZBTB46 defines and regulates ILC3s that protect the intestine. Nature 609, 159–165 (2022).
Salvermoser, J. et al. Clec9a-mediated ablation of conventional dendritic cells suggests a lymphoid path to generating dendritic cells in vivo. Front. Immunol. 9, 699 (2018).
Maier, B. et al. A conserved dendritic-cell regulatory program limits antitumour immunity. Nature 580, 257–262 (2020).
Brown, C. C. et al. Transcriptional basis of mouse and human dendritic cell heterogeneity. Cell 179, 846–863 (2019).
Cheng, S. et al. A pan-cancer single-cell transcriptional atlas of tumor infiltrating myeloid cells. Cell 184, 792–809 (2021).
Weiler, P., Lange, M., Klein, M., Pe’er, D. & Theis, F. CellRank 2: unified fate mapping in multiview single-cell data. Nat. Methods 21, 1196–1205 (2024).
Setty, M. et al. Characterization of cell fate probabilities in single-cell data with Palantir. Nat. Biotechnol. 37, 451–460 (2019).
Cugurra, A. et al. Skull and vertebral bone marrow are myeloid cell reservoirs for the meninges and CNS parenchyma. Science https://doi.org/10.1126/science.abf7844 (2021).
Wu, S. Y., Fu, T., Jiang, Y. Z. & Shao, Z. M. Natural killer cells in cancer biology and therapy. Mol. Cancer 19, 120 (2020).
Spector, R., Robert Snodgrass, S. & Johanson, C. E. A balanced view of the cerebrospinal fluid composition and functions: focus on adult humans. Exp. Neurol. 273, 57–68 (2015).
Oppenheim, H. Über Hirnsymptome bei Carcinomatose ohne nachweisbare Veränderungen im Gehirn. Charité Annal. 13, 335–344 (1888).
Schwarz, E. & Bertels, A. Über “Meningitis” carcinomatosa. Deut. Z. Nervenheilkd. 42, 85–94 (1911).
Whiteley, A. E. et al. Breast cancer exploits neural signaling pathways for bone-to-meninges metastasis. Science 384, eadh5548 (2024).
Gocher, A. M., Workman, C. J. & Vignali, D. A. A. Interferon-γ: teammate or opponent in the tumour microenvironment? Nat. Rev. Immunol. 22, 158–172 (2022).
Fernandez-Castaneda, A. et al. Mild respiratory SARS-CoV-2 infection can cause multi-lineage cellular dysregulation and myelin loss in the brain. Preprint at bioRxiv https://doi.org/10.1101/2022.01.07.475453 (2022).
Kaplan, D. H. et al. Demonstration of an interferon γ-dependent tumor surveillance system in immunocompetent mice. Proc. Natl Acad. Sci. USA 95, 7556–7561 (1998).
Dutta, T., Spence, A. & Lampson, L. A. Robust ability of IFN-γ to upregulate class II MHC antigen expression in tumor bearing rat brains. J. Neurooncol. 64, 31–44 (2003).
Propper, D. J. et al. Low-dose IFN-γ induces tumor MHC expression in metastatic malignant melanoma. Clin. Cancer Res. 9, 84–92 (2003).
Naidoo, J. et al. Pembrolizumab for patients with leptomeningeal metastasis from solid tumors: efficacy, safety, and cerebrospinal fluid biomarkers. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-002473 (2021).
Brastianos, P. K. et al. Single-arm, open-label phase 2 trial of pembrolizumab in patients with leptomeningeal carcinomatosis. Nat. Med. 26, 1280–1284 (2020).
Brastianos, P. K. et al. Phase II study of ipilimumab and nivolumab in leptomeningeal carcinomatosis. Nat. Commun. 12, 5954 (2021).
Myers, J. A. & Miller, J. S. Exploring the NK cell platform for cancer immunotherapy. Nat. Rev. Clin. Oncol. 18, 85–100 (2021).
Murphy, T. L. & Murphy, K. M. Dendritic cells in cancer immunology. Cell. Mol. Immunol. 19, 3–13 (2022).
Azizi, E. et al. Single-cell map of diverse immune phenotypes in the breast tumor microenvironment. Cell 174, 1293–1308 (2018).
Chan, J. M. et al. Signatures of plasticity, metastasis, and immunosuppression in an atlas of human small cell lung cancer. Cancer Cell 39, 1479–1496 (2021).
Gayoso, A., Shor, J., Carr, A. J., Sharma, R., Pe’er, D. DoubletDetection (version v3.0). Zenodo https://doi.org/10.5281/zenodo.2678041 (2020).
Hie, B., Bryson, B. & Berger, B. Efficient integration of heterogeneous single-cell transcriptomes using Scanorama. Nat. Biotechnol. 37, 685–691 (2019).
Traag, V. A., Waltman, L. & van Eck, N. J. From Louvain to Leiden: guaranteeing well-connected communities. Sci. Rep. 9, 5233 (2019).
Chan, J. M. et al. Lineage plasticity in prostate cancer depends on JAK/STAT inflammatory signaling. Science 377, 1180–1191 (2022).
van Dijk, D. et al. Recovering gene interactions from single-cell data using data diffusion. Cell 174, 716–729 (2018).
Wolf, F. A., Angerer, P. & Theis, F. J. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 19, 15 (2018).
Bergen, V., Lange, M., Peidli, S., Wolf, F. A. & Theis, F. J. Generalizing RNA velocity to transient cell states through dynamical modeling. Nat. Biotechnol. 38, 1408–1414 (2020).
Mombaerts, P. et al. RAG-1-deficient mice have no mature B and T lymphocytes. Cell 68, 869–877 (1992).
Jung, S. et al. Analysis of fractalkine receptor CX3CR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol. Cell. Biol. 20, 4106–4114 (2000).
Saederup, N. et al. Selective chemokine receptor usage by central nervous system myeloid cells in CCR2-red fluorescent protein knock-in mice. PLoS ONE 5, e13693 (2010).
Lee, S. H. et al. Identifying the initiating events of anti-Listeria responses using mice with conditional loss of IFN-γ receptor subunit 1 (IFNGR1). J. Immunol. 191, 4223–4234 (2013).
Hennet, T., Hagen, F. K., Tabak, L. A. & Marth, J. D. T-cell-specific deletion of a polypeptide N-acetylgalactosaminyl-transferase gene by site-directed recombination. Proc. Natl Acad. Sci. USA 92, 12070–12074 (1995).
Clausen, B. E., Burkhardt, C., Reith, W., Renkawitz, R. & Forster, I. Conditional gene targeting in macrophages and granulocytes using LysMcre mice. Transgenic Res. 8, 265–277 (1999).
Narni-Mancinelli, E. et al. Fate mapping analysis of lymphoid cells expressing the NKp46 cell surface receptor. Proc. Natl Acad. Sci. USA 108, 18324–18329 (2011).
Buch, T. et al. A Cre-inducible diphtheria toxin receptor mediates cell lineage ablation after toxin administration. Nat. Methods 2, 419–426 (2005).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Ferris, S. T. et al. cDC1 prime and are licensed by CD4+ T cells to induce anti-tumour immunity. Nature 584, 624–629 (2020).
Remsik, J. et al. TGF-β regulates Sca-1 expression and plasticity of pre-neoplastic mammary epithelial stem cells. Sci. Rep. 10, 11396 (2020).
Remsik, J. et al. Leptomeningeal metastatic cells adopt two phenotypic states. Cancer Rep. 5, e1236 (2022).
Putri, G. H., Anders, S., Pyl, P. T., Pimanda, J. E. & Zanini, F. Analysing high-throughput sequencing data in Python with HTSeq 2.0. Bioinformatics 38, 2943–2945 (2022).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Roelli, P., Flynn, B., Gui, G. Hoohm/CITE-seq-Count: 1.4.2 (version 1.4.2). Zenodo https://doi.org/10.5281/zenodo.2590196 (2019).
Gayoso, A. et al. Joint probabilistic modeling of single-cell multi-omic data with totalVI. Nat. Methods 18, 272–282 (2021).
Gulati, G. S. et al. Single-cell transcriptional diversity is a hallmark of developmental potential. Science 367, 405–411 (2020).
Reuter, B., Weber, M., Fackeldey, K., Roblitz, S. & Garcia, M. E. Generalized Markov state modeling method for nonequilibrium biomolecular dynamics: exemplified on amyloid beta conformational dynamics driven by an oscillating electric field. J. Chem. Theory Comput. 14, 3579–3594 (2018).
Kolabas, Z. I. et al. Distinct molecular profiles of skull bone marrow in health and neurological disorders. Cell 186, 3706–3725 (2023).
Remsik, J. et al. Plasticity and intratumoral heterogeneity of cell surface antigen expression in breast cancer. Br. J. Cancer https://doi.org/10.1038/bjc.2017.497 (2018).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Acknowledgements
We thank the patients and families who inspired this work and donated clinical samples enabling this research; A. Skakodub, R. Estrera and I. Osei-Gyening for management of the human CSF bank; C. Burdziak, L. DeAngelis, M. Freret, A. Schietinger, R. Sharma, T. Walle and J. Wolchok for discussions; M. Alghader, C. Derderian, S. Piedrafita-Ortiz, R. Uwayesu and D. Guber for technical assistance; I. Masilionas, B. Meškauskaitė and O. Chaudhary for help with wet laboratory single-cell processing; Y. Miranda for veterinary assistance; N. Fan, E. Chan and E. Rosiek for assistance with histology and image quantification; Y. F. Parisotto and L. Fisher for maintenance of Clec9a-cre and lsl-DTR colonies; S. Grassmann for maintenance of Nkp46-cre and Ifng-flox/flox colonies; and P. Weiler for guidance with statistical analyses. Clec9a-cre, Ifng-flox/flox and Nkp46-cre transgenic lines were provided by C. Reis e Sousa, A. Rudensky and E. Vivier, respectively. Financial support for this research was provided to A.B. by the Damon Runyon Cancer Research Foundation, the Pershing Square Sohn Cancer Research Alliance, the Alan and Sandra Gerry Metastasis and Tumor Ecosystems Center and NCI R01CA245499. J.R. was supported by the American Brain Tumor Association Basic Research Fellowship, the Terri Brodeur Breast Cancer Foundation, the Fiona and Stanley Druckenmiller Center for Lung Cancer Research and the VIB Core Funding. All investigations were supported by NCI P30 CA008748 (Cancer Center Support Grant). This publication is part of the HTAN (Human Tumor Atlas Network) Consortium paper package. A list of HTAN members is available online (https://humantumoratlas.org/htan-authors/).
Author information
Authors and Affiliations
Contributions
J.R. and A.B. conceived and designed the study, acquired funding, wrote and revised the manuscript. J.R. generated metastatic cell lines, developed the methodology, performed the experiments, analysed and curated the data. X.T., M.J.L., D.I., J.S., R.E., C.T. and H.W. assisted with experiments and replicated critical experiments. J.R. and R.Z.K. performed computational analysis under D.P.’s supervision. A.M.O. assisted with immunofluorescence staining and image analysis. K.C., J.A.W. and U.T.S. assisted with clinical annotations and human CSF collection. T.A.B. reviewed cytospin staining. R.C. supervised single-cell sequencing. J.C.S. and C.B. provided transgenic mouse lines. All of the authors approved the final manuscript. A.B. supervised the study.
Corresponding author
Ethics declarations
Competing interests
J.R. and A.B. are listed as inventors on provisional patent applications based on this study, filed by MSKCC. A.B. holds an unpaid position on the scientific advisory board for Evren Scientific and is listed as an inventor on the following patents: 62/258,044, 10/413,522 and 63/052,139. D.P. is on the scientific advisory board of Insitro. The other authors declare no competing interests.
Peer review
Peer review information
Nature thanks Michael Taylor and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Targeted proteomics with proximity extension assay of inflammatory mediators in human CSF.
(a) Targeted proteomic analysis of 92 inflammatory mediators in CSF of breast cancer patients without and with LM by proximity extension assay (multiple t-tests). (b) Targeted proteomic analysis of 92 inflammatory mediators in CSF of lung cancer patients without and with LM by proximity extension assay (multiple t-tests). (c) Targeted proteomic analysis of 92 inflammatory mediators in CSF of melanoma patients without and with LM by proximity extension assay (multiple t-tests). (d) Overlap of inflammatory mediators significantly enriched in CSF of LM+ patients, plotted per primary cancer type (Venn diagram, top panel). Overview of 15 proteins enriched in CSF from LM+ patients and all three cancer types. (e) Scatter plots show numbers of CSF leukocytes, lymphocytes, myeloid cells, neutrophils, and cancer cells for patients with available clinical differential counts; low (n = 25) and high (n = 28) levels of CSF IFN-γ, related to Fig. 1e. (f) Levels of CSF IFN-γ in cancer patients with or without LM from wide array of solid tumours, as determined with ELISA (LM- n = 50, LM+ n = 107). Patients with undetectable (u.d.) IFN-γ levels and patients with high outliers were used for correlative analyses in panels g and h. (g) Kaplan-Meier plot showing post-LM diagnosis survival in relation to CSF IFN-γ levels, related to panel f; logrank test. (h) Scatter plots show numbers of CSF leukocytes, lymphocytes, myeloid cells, neutrophils, and cancer cells for patients with available clinical differential counts; u.d. (n = 28) and high (n = 16) levels of CSF IFN-γ, related to panel g.
Extended Data Fig. 2 Immunocompetent mouse models of leptomeningeal metastasis.
(a) Overview of cancer cells lines used and generated in this study. (b) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3), LeptoM (purple, n = 3), and BrM1 (orange, n = 3) LLC cells. Retrieved from NCBI GEO GSE83132. (c) Kaplan-Meier plot showing survival of C57Bl/6-Tyrc-2 animals overexpressing Egfp in the leptomeninges after delivery of LLC LeptoM cells into cisterna magna (related to Fig. 3c). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of LLC LeptoM cells (scale bar = 100 μm). mOS - median overall survival. (d) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3) and newly established LeptoM (purple, n = 4), and BrM2 (orange, n = 3) B16 cells. (e) Kaplan-Meier plot showing survival of C57Bl/6 animals overexpressing Egfp in the leptomeninges after delivery of B16 LeptoM cells into cisterna magna (related to Extended Data Fig. 9f). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of B16 LeptoM cells (scale bar = 100 μm). mOS - median overall survival. (f) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3) and newly established LeptoM (purple, n = 3), and BrM2 (orange, n = 3) Yumm5.2 cells. (g) Kaplan-Meier plot showing survival of C57Bl/6 and C57Bl/6-Tyrc-2 animals overexpressing Egfp in the leptomeninges after delivery of Yumm5.2 LeptoM cells into cisterna magna (related to Extended Data Fig. 9l). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of Yumm5.2 LeptoM cells (scale bar = 100 μm). mOS - median overall survival. (h) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3) and newly established LeptoM (purple, n = 5), and BrM2 (orange, n = 3) E0771 cells. (i) Kaplan-Meier plot showing survival of C57Bl/6-Tyrc-2 animals overexpressing Egfp in the leptomeninges after delivery of E0771 LeptoM cells into cisterna magna (related to Extended Data Fig. 9d). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of E0771 LeptoM cells (scale bar = 100 μm). mOS - median overall survival. (j) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3) and newly established LeptoM (purple, n = 3), and BrM2 (orange, n = 3) EMT6 cells. (k) Kaplan-Meier plot showing survival of BALB/c animals overexpressing Egfp in the leptomeninges after delivery of EMT6 LeptoM cells into cisterna magna (related to Extended Data Fig. 9h). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of EMT6 LeptoM cells (scale bar = 100 μm). mOS - median overall survival. (l) Principal component analysis (PCA) of in vitro transcriptome of Parental (grey, n = 3) and newly established LeptoM (purple, n = 3), and BrM2 (orange, n = 3) 4T1 cells. (m) Kaplan-Meier plot showing survival of BALB/c animals overexpressing Egfp in the leptomeninges after delivery of 4T1 LeptoM cells into cisterna magna (related to Extended Data Fig. 9j). Representative brain tissue sections stained with H&E showing colonization of leptomeninges after intracardiac delivery of 4T1 LeptoM cells (scale bar = 100 μm). mOS - median overall survival.
Extended Data Fig. 3 Interaction between leptomeningeal metastasis and systemic immunity.
(a) In vivo radiance of subcutaneous LLC LeptoM tumours two weeks after injection of leptomeningeal tumour from the same subline. Quantification shows extracranial radiance at four weeks. (b) In vivo radiance of leptomeningeal LLC LeptoM tumours in the presence of subcutaneous tumour from the same subline. Extracranial tumours were injected two weeks before intracisternal implantation, and quantification shows cranial radiance after two weeks. Related to panel N. (c) In vivo radiance of mammary E0771 LeptoM tumour two weeks after injection of leptomeningeal tumour from the same subline. Quantification shows extracranial radiance at four weeks. (d) In vivo radiance of leptomeningeal E0771 LeptoM tumours in the presence of mammary tumour from the same subline. Extracranial tumours were injected two weeks before intracisternal implantation, and quantification shows cranial radiance after two weeks. Related to panel P. (e) Proportion of major leptomeningeal immune cell types in control and LLC LeptoM-bearing mice. Cells were injected subcutaneously and/or intracisternally. Related to panel N. (f) Proportion of major leptomeningeal immune cell types in control and LLC LeptoM-bearing mice. Cells were injected into lungs and/or intracisternally. (g) Proportion of major leptomeningeal immune cell types in control and E0771 LeptoM-bearing mice. Cells injected into mammary fat pad and/or intracisternally. Related to panel P. (h) Proportion of major leptomeningeal and lung immune cell types in control and E0771 LeptoM-bearing mice. Cells were injected intracardially (multiple t-tests; column numbers for comparison are shown below the group sizes, B – B cells, T – T cells, MM – monocytes & macrophages, Neu – neutrophils, NK – NK cells, cDC – conventional dendritic cells, pDC – plasmacytoid dendritic cells).
Extended Data Fig. 4 IFN-γ production and response in leptomeninges.
(a) Proportion of T cells (CD3+CD4+CD8- vs. CD3+CD4-CD8+) and NK cells (CD3-Nk1.1+) expressing IFN-γ in cells isolated from vehicle- or B16, E0771, and LLC LeptoM-injected mice, determined with flow cytometry. (b) Expression of IFNG gene in mouse (left) and human (right) single-cell datasets. (c) Abundance of phosphorylated STAT1 (pSTAT1) in leptomeningeal dendritic cells (MHC II+ CD11c+), monocyte-macrophages (CD11b+Ly6C+ and CD11b+F4/80+), T cells (CD3+), and NK cells (Nk1.1+), as a proxy for IFN-γ pathway activation in vehicle- and LLC LeptoM-injected mice, determined with flow cytometry. (d) Gene scores of IFN-γ-associated gene set, identified in this study and projected onto human CSF single-cell sequencing data from Fig. 1b. See Methods for details. (e) Gene scores of Hallmark Interferon Gamma Response gene set, projected onto human CSF single-cell sequencing data from Fig. 1b. (f) Proportion of major leptomeningeal immune cell types in the leptomeninges of LLC LeptoM-bearing mice depleted for NK cells and T cells two weeks after injection and continuous depletion (multiple t-tests). (g) Proportion of major leptomeningeal immune cell types in the leptomeninges of LLC LeptoM-bearing T cellwt and T cellΔIfng mice (multiple t-tests). (h) Proportion of major leptomeningeal immune cell types in the leptomeninges of LLC LeptoM-bearing NK cellwt and NK cellΔIfng mice (multiple t-tests). (i) Abundance of phosphorylated STAT1 (pSTAT1) in leptomeningeal dendritic cells (MHC II+ CD11c+) in LLC LeptoM-bearing mice depleted for NK cells and T cells, in T cellwt and T cellΔIfng mice, and in NK cellwt and NK cellΔIfng mice two weeks after injection. (j) Dendritic cell counts in leptomeninges from LLC LeptoM-bearing mice depleted for NK cells and T cells, in T cellwt and T cellΔIfng mice, and in NK cellwt and NK cellΔIfng mice two weeks after injection. (k) NK cell counts in leptomeninges from LLC LeptoM-bearing mice depleted for NK cells and T cells, in T cellwt and T cellΔIfng mice, and in NK cellwt and NK cellΔIfng mice two weeks after injection. (B – B cells, T – T cells, MM – monocytes & macrophages, Neu – neutrophils, NK – NK cells, DC – dendritic cells).
Extended Data Fig. 5 Leptomeningeal tumour growth in Ifng- and Ifngr1-deficient animals.
(a) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of E0771 LeptoM cells delivered intracisternally into C57BL/6 Ifng-proficient and -deficient animals, quantified two weeks after injection. (b) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of LLC LeptoM cells delivered intracisternally into C57BL/6 Ifng-proficient and -deficient animals, quantified two weeks after injection. (c) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of E0771 LeptoM cells delivered intracisternally into C57BL/6 Ifngr1-proficient and -deficient animals, quantified two weeks after injection. (d) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of LLC LeptoM cells delivered intracisternally into C57BL/6 Ifngr1-proficient and -deficient animals, quantified two weeks after injection. (e) Proportion of major leptomeningeal immune cell types in naïve and LLC LeptoM-bearing C57BL/6 Ifngr1-proficient and -deficient animals two weeks after vehicle or intracisternal cancer cell injection, determined with flow cytometry (multiple t-tests; B – B cells, T – T cells, MM – monocytes & macrophages, Neu – neutrophils, NK – NK cells, cDC – conventional dendritic cells, pDC – plasmacytoid dendritic cells).
Extended Data Fig. 6 Extracranial tumour growth in Ifng- and Ifngr1-deficient animals.
(a) Volumes of intradermal B16 LeptoM flank tumours in C57BL/6 Ifng-proficient and -deficient animals, quantified two weeks after injection. (b) Volumes of mammary fat pad E0771 LeptoM tumours in C57BL/6 Ifng-proficient and -deficient animals, quantified four weeks after injection. (c) Volumes of subcutaneous LLC LeptoM flank tumours in C57BL/6 Ifng-proficient and -deficient animals, quantified three weeks after injection. (d) Volumes of intradermal B16 LeptoM flank tumours in C57BL/6 Ifngr1-proficient and -deficient animals, quantified two weeks after injection. (e) Volumes of mammary fat pad E0771 LeptoM tumours in C57BL/6 Ifngr1-proficient and -deficient animals, quantified four weeks after injection. (f) Volumes of subcutaneous LLC LeptoM flank tumours in C57BL/6 Ifngr1-proficient and -deficient animals, quantified three weeks after injection. (g) Cranial and extracranial in vivo radiance of non-metastatic E0771 Parental cells injected intracardially into C57BL/6 Ifngr1-proficient and -deficient animals two weeks after injection. Notably, one Ifngr1-deficient animal developed histologically confirmed periventricular lesion with cancer cells spread to the contralateral ventricle. (h) Incidence of overt radiographic metastases in cranial (brain), thoracic (lung), and pelvic (femoral bone) regions of C57BL/6 Ifngr1-proficient and -deficient animals from Extended Data Fig. 6g. (i) Cranial and extracranial in vivo radiance of brain-metastatic E0771 BrM2 cells injected intracardially into C57BL/6 Ifngr1-proficient and -deficient animals two weeks after injection. (j) Incidence of overt radiographic metastases in cranial (brain), thoracic (lung), and pelvic (femoral bone) regions of C57BL/6 Ifngr1-proficient and -deficient animals from Extended Data Fig. 6i.
Extended Data Fig. 7 Cancer-intrinsic IFN-γ signalling is dispensable for tumour growth in leptomeninges.
(a) In vitro induction of MHC class I in control (sgLacZ) and two Ifngr2-deficient E0771 LeptoM clones with recombinant IFN-γ. Data pooled from three independent experiments. (b) In vitro induction of MHC class I in control (sgLacZ) and two Ifngr2-deficient LLC LeptoM clones with recombinant IFN-γ. Data pooled from three independent experiments. (c) In vitro induction of MHC class I in control (sgLacZ) and two Ifngr2-deficient B16 LeptoM clones with recombinant IFN-γ. Data pooled from three independent experiments. (d) In vitro proliferation of control (sgLacZ) and two Ifngr2-deficient E0771 LeptoM clones exposed to recombinant IFN-γ. Data pooled from three independent experiments. (e) In vitro proliferation of control (sgLacZ) and two Ifngr2-deficient LLC LeptoM clones exposed to recombinant IFN-γ. Data pooled from three independent experiments. (f) In vitro proliferation of control (sgLacZ) and two Ifngr2-deficient B16 LeptoM clones exposed to recombinant IFN-γ. Data pooled from three independent experiments. (g) In vivo radiance of control (sgLacZ) and two Ifngr2-deficient E0771 LeptoM clones delivered intracisternally into C57Bl/6-Tyrc-2 animals, quantified three weeks after injection in one in vivo experiment. (h) In vivo radiance of control (sgLacZ) and two Ifngr2-deficient LLC LeptoM clones delivered intracisternally into C57Bl/6-Tyrc-2 animals, quantified two weeks after injection in one in vivo experiment.
Extended Data Fig. 8 Leptomeningeal IFN-γ-mediated tumour growth suppression is driven by the microenvironment.
(a) In vitro induction of MHC class I in E0771 LeptoM cells with recombinant IFN-γ. Data pooled from three independent experiments. (b) In vitro induction of MHC class I in LLC LeptoM cells with recombinant IFN-γ. Data pooled from three independent experiments. (c) In vitro induction of MHC class I in B16 LeptoM cells with recombinant IFN-γ. Data pooled from three independent experiments. (d) In vitro proliferation of E0771 LeptoM cells exposed to recombinant IFN-γ. Data pooled from three independent experiments. (e) In vitro proliferation of LLC LeptoM cells exposed to recombinant IFN-γ. Data pooled from three independent experiments. (f) In vitro proliferation of B16 LeptoM cells exposed to recombinant IFN-γ. Data pooled from three independent experiments. (g) In vivo tumour growth of E0771 LeptoM cells in C57Bl/6-Tyrc-2 animals injected weekly with vehicle or two doses of recombinant IFN-γ, as a function of radiance. (h) In vivo tumour growth of LLC LeptoM cells in C57Bl/6-Tyrc-2 animals injected weekly with vehicle or two doses of recombinant IFN-γ, as a function of radiance. (i) In vivo tumour growth of B16 LeptoM cells in C57Bl/6 animals injected weekly with vehicle or two doses of recombinant IFN-γ, as a function of radiance. (j) In vivo tumour growth of LLC LeptoM cells in C57Bl/6-Tyrc-2 animals injected weekly with heat-inactivated vehicle (PBS) or heat-inactivated recombinant IFN-γ, as a function of radiance in one in vivo experiment.
Extended Data Fig. 9 Leptomeninges-specific overexpression of IFN-γ extends survival of LeptoM cells-bearing animals.
(a) Schematic showing experimental strategy of leptomeningeal Egfp or Ifng overexpression, used for functional experiments in this study. (b) Levels of IFN-γ in the CSF collected from naïve C57Bl/6 and BALB/c animals overexpressing Egfp or Ifng in the leptomeninges, detected by cytometric bead array. (c) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of E0771 LeptoM cells delivered intracisternally into C57Bl/6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges, quantified two weeks after injection. (d) Kaplan-Meier plot showing survival of E0771 LeptoM-bearing C57Bl/6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (e) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates brain surface area covered with pigmented B16 LeptoM cells delivered intracisternally into C57BL/6 animals overexpressing Egfp or Ifng in the leptomeninges, quantified two weeks after injection. (f) Kaplan-Meier plot showing survival of B16 LeptoM-bearing C57Bl/6 animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (g) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of EMT6 LeptoM cells delivered intracisternally into BALB/c animals overexpressing Egfp or Ifng in the leptomeninges, quantified two weeks after injection. (h) Kaplan-Meier plot showing survival of EMT6 LeptoM-bearing BALB/c animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (i) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of 4T1 LeptoM cells delivered intracisternally into BALB/c animals overexpressing Egfp or Ifng in the leptomeninges, quantified one week after injection. (j) Kaplan-Meier plot showing survival of 4T1 LeptoM-bearing BALB/c animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (k) Representative leptomeningeal tissue sections stained with H&E (scale bar = 100 μm). Box plot illustrates in vivo radiance of Yumm5.2 LeptoM cells delivered intracisternally into C57Bl6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges, quantified three weeks after injection. (l) Kaplan-Meier plot showing survival of Yumm5.2 LeptoM-bearing C57Bl/6 and C57Bl/6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges (logrank test).
Extended Data Fig. 10 Leptomeningeal IFN-γ does not affect morphology of brain parenchyma but reduces oligodendrocyte numbers in corpus callosum.
(a) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained with Luxol Fast Blue (n = 4 per group, 3 months after AAV introduction, scale bar = 500 μm). (b) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for astrocyte activation marker GFAP (n = 4 per group, 3 months after AAV introduction, scale bar = 100 μm) and quantification of GFAP+ periventricular layer thickness. (c) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for microglia marker Iba1 (n = 4 per group, 3 months after AAV introduction, scale bar = 100 μm) and quantification of Iba1+ microglia in cortical layers 3 and 4. (d) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for neural progenitor marker DCX (n = 4 per group, 3 months after AAV introduction, scale bar = 100 μm) and quantification of DCX fluorescence in the hippocampus. (e) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for myelinization marker MBP (n = 4 per group, 3 months after AAV introduction, scale bar = 200 μm) and quantification of overage MBP+ tract area per hemisphere. (f) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for marker of mature neurons NeuN (n = 4 per group, 3 months after AAV introduction, scale bar = 200 μm) and quantification of NeuN+ mature neurons per FOV in cortical layers 1–4 (left) and 5-6 (right). See outline in panel F. (g) Quantification of leptomeningeal leukocytes. (h) Flow cytometric analysis of major leptomeningeal immune cell types (multiple t-tests; B – B cells, T – T cells, MM – monocytes & macrophages, Neu – neutrophils, NK – NK cells, cDC – conventional dendritic cells, pDC – plasmacytoid dendritic cells). (i) Representative images of corpus callosum sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for marker of oligodendrocytes Olig2 (n = 4 per group, 3 months after AAV introduction, scale bar = 100 μm), and quantification of Olig2+ oligodendrocytes per FOV in corpus callosum. (j) Representative images of brain tissue sections from naïve C57Bl/6 animals overexpressing Egfp or Ifng stained for markers of oligodendrocytes Olig2 and CNPase (n = 4 per group, 3 months after AAV introduction, scale bar = 100 μm) and quantification of Olig2+ oligodendrocytes per FOV in cortical and subcortical regions, and CNPase+ oligodendrocytes per FOV in cortical and subcortical regions (corresponding regions are marked in the representative images).
Extended Data Fig. 11 Leptomeningeal IFN-γ does not require adaptive immune system and signals through conventional dendritic cells to suppress metastatic outgrowth.
(a) In vivo radiance of E0771 LeptoM cells delivered intracisternally into NSG animals overexpressing Egfp or Ifng in the leptomeninges, quantified two weeks after injection. (NSG - non-obese, diabetic, severe combined immunodeficient, Il2rgnull). (b) Kaplan-Meier plot showing survival of LLC LeptoM-bearing NSG animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (c) Kaplan-Meier plot showing survival of E0771 LeptoM-bearing NSG animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (d) In vivo radiance of E0771 LeptoM cells delivered intracisternally into Rag1-deficient animals overexpressing Egfp or Ifng in the leptomeninges, quantified two weeks after injection. (NSG - non-obese, diabetic, severe combined immunodeficient, Il2rgnull). (e) Kaplan-Meier plot showing survival of LLC LeptoM-bearing Rag1-deficient animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (f) Kaplan-Meier plot showing survival of E0771 LeptoM-bearing Rag1-deficient animals overexpressing Egfp or Ifng in the leptomeninges (logrank test). (g) Representative images of dendritic cell marker CD11c in leptomeningeal cancer plaques in wild-type (WT) and Zbtb46-DTR bone marrow chimeras, treated with diphtheria toxin (DTx). Mice were injected with LLC LeptoM cells (scale bar = 20 μm). (h) Representative images of dendritic cell marker CD11c in spleen of wild-type (WT) and Zbtb46-DTR bone marrow chimeras, treated with diphtheria toxin (DTx). Mice were injected with LLC LeptoM cells (scale bar = 20 μm). (i) Quantification of systemic cDC depletion in images from panel B. (j) In vivo radiance of LLC LeptoM cells delivered subcutaneously into Zbtb46-DTR bone marrow chimeras. Recipient mice were on C57Bl6-Tyrc-2 background, bioluminescence was measured two weeks after cancer cell injection after continuous administration of vehicle (saline) or diphtheria toxin. (k) In vivo radiance of E0771 LeptoM cells delivered intracisternally into Clec9acre and Clec9acreRosa26lsl-DTR mice after administration of diphtheria toxin two weeks after injection. (l) In vivo radiance of LLC LeptoM cells delivered subcutaneously into Clec9acre and Clec9acreRosa26lsl-DTR mice after administration of diphtheria toxin two weeks after injection. (m) Proportion of major leptomeningeal immune cell types in naïve and LLC LeptoM-bearing Clec9acre and Clec9acreRosa26lsl-DTR animals two weeks after intracisternal cancer cell injection and continuous administration of diphtheria toxin, determined with flow cytometry (multiple t-tests; B – B cells, T – T cells, MM – monocytes & macrophages, Neu – neutrophils, NK – NK cells, cDC – conventional dendritic cells, pDC – plasmacytoid dendritic cells).
Extended Data Fig. 12 Trajectory analysis of leptomeningeal dendritic cells.
(a) tSNE maps showing abundance of captured dendritic cell types in naïve and metastasis-bearing, Egfp- or Ifng-overexpressing mice (total of n = 7,566 cells pooled from 4 conditions and n = 6 animals per group). (b) Proportion of dendritic cell subtypes per condition. (c) Counts of dendritic cell subtypes per condition. (d) tSNE projection of dendritic cell surface markers detected with CITE-seq. CD11c - pan-DC marker; Xcr1 - cDC1 marker; CD11b - cDC2 marker; B220 - pDC marker. (e) Bivariate plot showing distribution of cell surface Xcr1 and CD11b in leptomeningeal dendritic cell subsets, as detected with CITE-seq. (f) tSNE projection of 2,575 mouse leptomeningeal DCs subsetted for trajectory analysis. Cells are from Egfp-overexpressing, naïve and cancer-bearing mice, and the plots are coloured based on cell type and condition. See Methods for further details. (g) tSNE projection of CytoTRACE pseudotime, as determined with CellRank, suggesting that CCR7+ DCs are the terminal state within the subsetted cell population. (h) Terminal DC macrostates and computed macrostate membership for each cell, as predicted with CellRank and projected onto a tSNE. While cDC1 cells are restricted to cDC1 membership, cells from cDC2 cluster are gradually acquiring CCR7 + DC membership. (i) Palantir-computed diffusion pseudotime and CCR7 + DC maturation (branch) probability. Gene trends along this pseudotime axis are plotted in Fig. 4d. (j) Plots show Pearson correlation of pseudotime orderings in Palantir analysis for different parameters (waypoint samplings, number of principal components, and number of K-nearest neighbours) and all cells. DC trajectory analysis, performed as described in Methods, is not sensitive to fluctuations in these parameters.
Extended Data Fig. 13 Lineage tracing of leptomeningeal CCR7+ dendritic cells and characterization metastatic cancer cells in Egfp- and Ifng-overexpressing mice.
(a) Proportion of leptomeningeal, CD11c+ MHC II+ double-positive cDC in LLC LeptoM-bearing Ifngr1-proficient and deficient mice. (b) Proportion leptomeningeal, IL12-producing CCR7+ DCs in LLC LeptoM-bearing Ifngr1-proficient and deficient mice. (c) Proportion of mCherry+ CCR7+ DCs across various conditions. mCherry reports the expression of Xcr1 and shows that only a minority of CCR7+ DCs expressed mCherry and originated in cDC1 subtype. (d) Classification of leptomeningeal, CD11c+ MHC II+ double-positive cDC into immature (CCR7-) and mature (CCR7+) subtypes across various conditions. (e) Proportion of Xcr1+ CCR7+ DCs across various conditions, confirming lineage tracing from Extended Data Fig. 12. (f) Schematic shows experimental design of strategy used to determine the cranial or extracranial origin of leptomeningeal immune cells. CD45.2 mice were irradiated with their cranium shielded and infused with CD45.1 bone marrow that colonized the depleted bones. CD45.2+ immune cells would after recovery predominantly originate in the shielded cranial bone marrow, while CD45.1+ immune cells would arrive from peripheral sites. (g) Quantification of bone marrow reconstitution. Cells in femoral bone marrow (BM) and circulation are mainly CD45.1 + , while cells isolated from cranial BM are CD45.2. (h) Determination of cDC subtype origin. Unlike neutrophils that are mostly sourced from proximal cranial BM, cDC are colonizing leptomeninges in naïve and LLC LeptoM-bearing animals from peripheral BM. (i) Quantification of cancer cells captured in the mouse single-cell atlas (n = 3,718 keratin+ CD63+ cells isolated from n = 6 mice per group); related to Fig. 4j. (j) GSEApy analysis of top 15 Reactome 2022 pathways enriched in cancer cells isolated from Ifng-overexpressing animals and subsetted as described in Fig. 4j (DEG cut-off P < 0.01). (k) GSEApy analysis of top 15 Reactome 2022 pathways enriched in cancer cells isolated from Egfp-overexpressing animals and subsetted as described in Fig. 4j (DEG cut-off P < 0.01). (l) Quantification of cleaved Caspase 3-positive cells in cancer plaques and clusters, in the leptomeninges of Egfp- or Ifng-overexpressing animals injected with LLC LeptoM cells. (m) Quantification of cleaved Caspase 3-positive cells in cancer plaques and clusters, in the leptomeninges of Egfp- or Ifng-overexpressing animals injected with E0771 LeptoM cells. (n) Quantification of cleaved Caspase 3-positive cells in cancer plaques and clusters, in the leptomeninges of Egfp- or Ifng-overexpressing animals injected with B16 LeptoM cells.
Extended Data Fig. 14 NK cells are the downstream cytotoxic effectors of leptomeningeal IFN-γ.
(a) Kaplan-Meier plot showing survival of LLC LeptoM-bearing C57Bl/6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges, depleted with control polyclonal antibody (left graph) or antibody targeting asialo-GM1 (logrank test). (b) Kaplan-Meier plot showing survival of E0771 LeptoM-bearing C57Bl/6-Tyrc-2 animals overexpressing Egfp or Ifng in the leptomeninges, depleted with control polyclonal antibody (left graph) or antibody targeting asialo-GM1 (logrank test). (c) Kaplan-Meier plot showing survival of B16 LeptoM-bearing C57Bl/6 animals overexpressing Egfp or Ifng in the leptomeninges, depleted with control polyclonal antibody (left graph) or antibody targeting asialo-GM1 in one experiment (logrank test). (d) Efficiency of systemic asialo-GM1-targeting depletion of NK cells in naïve C57Bl/6 animals, quantified in leptomeninges with flow cytometry. (e) Efficiency of systemic asialo-GM1-targeting depletion of NK cells in naïve C57Bl/6 animals, quantified in spleen with flow cytometry. (f) Proportion of EdU+ NK cells in LLC LeptoM-bearing NK cellwt and NK cellΔIfngr1 animals two weeks after intracisternal cancer cell injection, determined with flow cytometry. (g) Proportion of EdU+ NK cells in LLC LeptoM-bearing C57BL/6 Ifngr1-proficient and -deficient animals two weeks after intracisternal cancer cell injection, determined with flow cytometry.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6.
Supplementary Tables
Supplementary Tables 1–6.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Remsik, J., Tong, X., Kunes, R.Z. et al. Interferon-γ orchestrates leptomeningeal anti-tumour response. Nature (2025). https://doi.org/10.1038/s41586-025-09012-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41586-025-09012-z
This article is cited by
-
CNS gatekeepers: IFNγ-activated dendritic cells control leptomeningeal metastasis
Cell Research (2025)